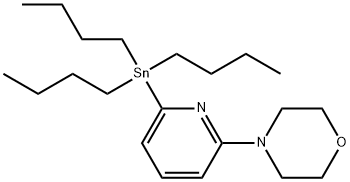
4-[6-(TRIBUTYLSTANNYL)-2-PYRIDINYL]MORPHOLINE synthesis
- Product Name:4-[6-(TRIBUTYLSTANNYL)-2-PYRIDINYL]MORPHOLINE
- CAS Number:869901-24-6
- Molecular formula:C21H38N2OSn
- Molecular Weight:453.25
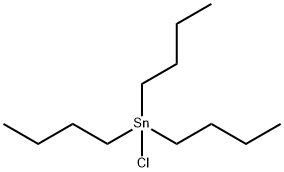
1461-22-9
213 suppliers
$10.00/10g
![4-[6-(TRIBUTYLSTANNYL)-2-PYRIDINYL]MORPHOLINE](/CAS/GIF/869901-24-6.gif)
869901-24-6
24 suppliers
inquiry
Yield:869901-24-6 7.2%
Reaction Conditions:
Stage #1: 4-(pyridin-2-yl)morpholinewith n-butyllithium;2-(N,N-dimethylamino)ethanol in hexane at -5 - 5; for 1.5 h;
Stage #2: tributyltin chloride in hexane at -78 - 20; for 2.5 h;
Stage #3: with water in hexane at 0;
Steps:
9.C.2
4-(6-Tributylstannanyl-pyridin-2-yl)-morpholineTo a solution of 2-dimethylaminoethanol (0.46 mL, 4.56 mmol) in hexane (7.0 mL, HPLC grade) cooled at -50C was added drop wise n-BuLJ (1.60 M1 5.70 mL, 9.12 mmol) under nitrogen atmosphere. After 30 min at O0C1 4-pyridin-2-yl-morpholine (0.25 g, 1.52 mmol) in hexane (2.0 mL) was added drop wise. After stirring the reaction mixture for 1 h at 0-50C, the reaction medium was cooled to -780C followed by drop wise addition of tributyl tin chloride (1.03 mL, 3.70 mmol). The resulting reaction mixture was stirred at -780C for 30 min and then allowed to stir at 0-50C for 2h. The reaction mixture was then allowed to come to room temperature. After the completion of reaction (TLC monitoring), the reaction mass was cooled to O0C and water was added slowly. The aqueous phase was extracted with diethyl ether (3x 20 mL). The combined organic layers were dried over anhydrous Na2SO4, and evaporated to dryness. The crude residue was purified over silica gel (230-400 M) using EtOAc-Hexane (2:98) to provide the compound as yellow oil (0.050 g, 7.20%). MS: 455 (M+H)+.
References:
WO2007/148093,2007,A1 Location in patent:Page/Page column 65-66