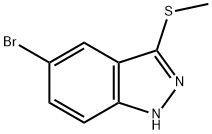
5-BroMo-3-(Methylthio)-1H-indazole synthesis
- Product Name:5-BroMo-3-(Methylthio)-1H-indazole
- CAS Number:201227-23-8
- Molecular formula:C8H7BrN2S
- Molecular Weight:243.12
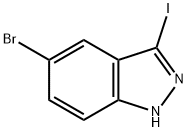
459133-66-5
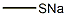
5188-07-8

201227-23-8
To a solution of 5-bromo-3-iodo-1H-indazole (3.7 g, 11.49 mmol) in dimethylsulfoxide (DMSO, 20 mL) was sequentially added an aqueous solution of 20% sodium methanethiol (MeSNa) (2.4 mL, 34.47 mmol) and cuprous iodide (CuI, 218 mg, 1.15 mmol). The resulting mixture was degassed and the reaction was heated at 120 °C for 3 h under nitrogen (N2) atmosphere. Upon completion of the reaction, it was cooled to room temperature and the reaction was quenched by the addition of water (50 mL), followed by extraction with ethyl acetate (50 mL x 3). The organic phases were combined, dried over anhydrous sodium sulfate and concentrated under reduced pressure. The residue was purified by fast column chromatography to afford the target product 5-bromo-3-(methylmercapto)-1H-indazole (2.5 g) as a yellow solid.

459133-66-5
157 suppliers
$10.00/250mg
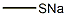
5188-07-8
296 suppliers
$13.00/5g

201227-23-8
22 suppliers
$142.00/100mg
Yield:201227-23-8 2.5 g
Reaction Conditions:
with copper(l) iodide in dimethyl sulfoxide at 120; for 3 h;Inert atmosphere;
Steps:
22.2 Preparation of 5-bromo -3-methylsulfanyl-lH-indazole
To a solution of 5-bromo-3-iodo-lH-indazo 2 (3.7 g, 11.49 mmol) in DMSO (20 mL) was added 20% aqueous MeSNa solution (2.4 mL, 34.47 mmol) and Cul (218 mg, 1.15 mmol) successively. The resulting mixture was degassed and charged with N2. After heating at 120 °C for 3 hours, the reaction was cooled down to room temperature. Water (50 mL) was added and the mixture was extracted with ethyl acetate (50 mL x 3). The combined organic layers were dried and concentrated under reduce pressure. The residue was purified by flash column to give 5-bromo-3-methylsulfanyl-lH-indazole (2.5 g) as a yellow solid.
References:
WO2014/90692,2014,A1 Location in patent:Page/Page column 56
53857-57-1
370 suppliers
$6.00/1g

201227-23-8
22 suppliers
$142.00/100mg