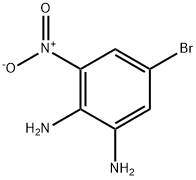
5-Bromo-3-nitro-benzene-1,2-diamine synthesis
- Product Name:5-Bromo-3-nitro-benzene-1,2-diamine
- CAS Number:84752-20-5
- Molecular formula:C6H6BrN3O2
- Molecular Weight:232.03
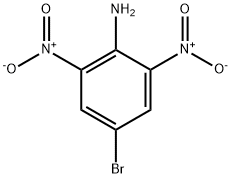
62554-90-9

84752-20-5
General procedure for the synthesis of 3-nitro-5-bromobenzene-1,2-diamine from 2,6-dinitro-4-bromoaniline: a mixture of 4-bromo-2,6-dinitroaniline (5.5 g, 21.2 mmol) and ammonium sulfide (10.5 mL, 21.2 mmol) in ethanol (150 mL) was heated and reacted for 1 hr at 90 °C. Thin layer chromatography (TLC) monitoring showed incomplete consumption of the feedstock. Ammonium sulfide (10.5 mL, 21.2 mmol) was added and the reaction was continued with stirring at 90 °C for 1 hour. Upon completion of the reaction, the mixture was concentrated and purified by silica gel column chromatography (eluent: petroleum ether/dichloromethane = 1:1) to afford the target compound 3-nitro-5-bromobenzene-1,2-diamine (2.5 g, 51% yield) as a red solid. Liquid chromatography-mass spectrometry (LCMS) analysis results: m/z 230, 232 ([M+H]+).

62554-90-9
45 suppliers
inquiry

84752-20-5
49 suppliers
$35.00/100mg
Yield:84752-20-5 51%
Reaction Conditions:
with diammonium sulfide in ethanol at 90; for 2 h;
Steps:
31.1 Step 1: 5-bromo-3-nitrobenzene-l,2-diamine
A mixture of 4-bromo-2,6-dinitroaniline (5.5 g, 21.2 mmol) and (NH4)2S (10.5 mL, 21.2 mmol) in ethanol (150 mL) was heated at 90 °C for 1 hr. TLC showed incomplete consumption of starting material. Additional (NH 2S (10.5 mL, 21.2 mmol) was added, and the reaction was stirred at 90 °C for 1 hr more. The mixture was concentrated and purified on silica gel (PE/DCM = 1 : 1) to give the title compound (2.5 g, 51%) as a red solid. LCMS: 230, 232 (M-H ).
References:
WO2016/168682,2016,A2 Location in patent:Paragraph 0288
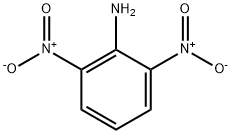
606-22-4
15 suppliers
$47.29/5g

84752-20-5
49 suppliers
$35.00/100mg