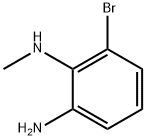
6-BroMo-N1-Methylbenzene-1,2-diaMine synthesis
- Product Name:6-BroMo-N1-Methylbenzene-1,2-diaMine
- CAS Number:1150102-47-8
- Molecular formula:C7H9BrN2
- Molecular Weight:201.06
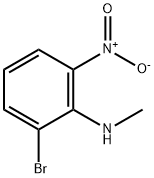
1004618-77-2
47 suppliers
inquiry

1150102-47-8
61 suppliers
$45.00/10mg
Yield: 99%
Reaction Conditions:
with iron;acetic acid in water;ethyl acetate at 50 - 80; for 4 h;
Steps:
2 Step 2-3-Bromo-N2-methyl-benzene-1,2-diamine
To a mixture of 2-bromo-N-methyl-6-nitro-aniline (23.0 g, 99.5 mmol) in EA (300 mL) and H2O (10 mL) was added AcOH (100 mL). The mixture was warmed to 50° C. Then Fe (22.2 g, 398 mmol) was added to the reaction mixture and the mixture was heated to 80° C. about 4 hours. On completion, the reaction mixture was filtered and concentrated in vacuo. The residue was diluted with water (100 mL) and extracted with EA (3×200 mL). The combined organic layers was dried over Na2SO4, filtered and concentrated in vacuo to give the title compound (20.0 g, 99% yield) as red oil. 1H NMR (400 MHz, DMSO-d6) δ 6.73-6.70 (m, 1H), 6.68-6.60 (m, 2H), 5.02 (s, 2H), 3.67 (s, 1H), 2.58 (s, 3H).
References:
Kymera Therapeutics, Inc.;Mainolfi, Nello;Ji, Nan;Kluge, Arthur F.;Weiss, Matthew M.;Zhang, Yi US2019/192668, 2019, A1 Location in patent:Paragraph 2881; 2883
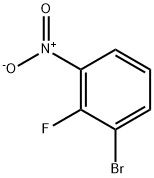
58534-94-4
187 suppliers
$10.00/1g

1150102-47-8
61 suppliers
$45.00/10mg