
Phosphinous acid, bis(1,1-dimethylethyl)-, (7S)-6,7-dihydro-2-phenyl-5H-cyclopenta[b]pyridin-7-yl ester (9CI) synthesis
- Product Name:Phosphinous acid, bis(1,1-dimethylethyl)-, (7S)-6,7-dihydro-2-phenyl-5H-cyclopenta[b]pyridin-7-yl ester (9CI)
- CAS Number:912277-31-7
- Molecular formula:C22H30NOP
- Molecular Weight:355.45
Yield:81.2 % ee
Reaction Conditions:
Stage #1: (S)-7-hydroxy-2-phenyl-6,7-dihydro-5H-<1>pyridinewith n-butyllithium in tetrahydrofuran;hexane at -78 - 20; for 0.5 h;Inert atmosphere;
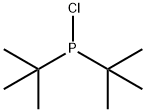
Stage #2: di(tert-butyl)chlorophosphine in tetrahydrofuran;hexane at -78 - 50;Inert atmosphere;
Steps:
General procedure: The ligand precursors (enantiomerically enriched secondary alcohols) were prepared according to known literature procedures like to the method disclosed in S. Kaiser et al., Angew. Chem. Int. Ed. 2006, 45, 5194- 5197 or in D. H. Woodmansee Chem. Sci 2010, 1, 72. The ligands and Iridium complexes were prepared by a modified procedure based on the same literature precedents: Procedure of ligand synthesis (under Ar): A solution of alcohol precursor in THF (0.25 mmol, in 5.0 mL THF) was cooled to -78 °C and n-BuLi (0.1 mL of a 2.5 M n-BuLi solution in hexane; 0.25 mmol; 1 eq.) was added dropwise to the continuously stirred solution. After completion of the addition the solution was allowed to warm to room temperature and was stirred at this temperature for further 30 min. The solution was cooled to -78 °C again and R2PCI (0.25 mmol, 1 eq.) was added to the continuously stirred solution. The mixture was allowed to warm to room temperature and subsequently heated to 50 °C and kept at this temperature overnight. The theoretical yield of ligand was calculated using 31P-NMR and the ligand was used for the next step without further purification.
References:
WO2019/185541,2019,A1 Location in patent:Page/Page column 25-26
![5H-Cyclopenta[b]pyridine, 6,7-dihydro-2-phenyl-, 1-oxide](/CAS/20210305/GIF/97360-41-3.gif)
97360-41-3
0 suppliers
inquiry
![Phosphinous acid, bis(1,1-dimethylethyl)-, (7S)-6,7-dihydro-2-phenyl-5H-cyclopenta[b]pyridin-7-yl ester (9CI)](/CAS/20210111/GIF/912277-31-7.gif)
912277-31-7
0 suppliers
inquiry
![5H-Cyclopenta[b]pyridine, 6,7-dihydro-2-phenyl-](/CAS/20210305/GIF/56396-63-5.gif)
56396-63-5
0 suppliers
inquiry
![Phosphinous acid, bis(1,1-dimethylethyl)-, (7S)-6,7-dihydro-2-phenyl-5H-cyclopenta[b]pyridin-7-yl ester (9CI)](/CAS/20210111/GIF/912277-31-7.gif)
912277-31-7
0 suppliers
inquiry

7095-09-2
1 suppliers
inquiry
![Phosphinous acid, bis(1,1-dimethylethyl)-, (7S)-6,7-dihydro-2-phenyl-5H-cyclopenta[b]pyridin-7-yl ester (9CI)](/CAS/20210111/GIF/912277-31-7.gif)
912277-31-7
0 suppliers
inquiry
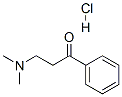
879-72-1
127 suppliers
$13.43/1gm:
![Phosphinous acid, bis(1,1-dimethylethyl)-, (7S)-6,7-dihydro-2-phenyl-5H-cyclopenta[b]pyridin-7-yl ester (9CI)](/CAS/20210111/GIF/912277-31-7.gif)
912277-31-7
0 suppliers
inquiry
![5H-Cyclopenta[b]pyridin-7-ol, 6,7-dihydro-2-phenyl-, (7S)-](/CAS/20210305/GIF/247185-53-1.gif)