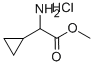
methyl-2-amino-2-cyclopropyla cetate hydrochloride synthesis
- Product Name:methyl-2-amino-2-cyclopropyla cetate hydrochloride
- CAS Number:535936-86-8
- Molecular formula:C6H11NO2.ClH
- Molecular Weight:165.619
10294-18-5

535936-86-8
General procedure for the synthesis of methyl 2-amino-2-cyclopropylacetate hydrochloride from DL-cyclopropylglycine: Thionyl chloride (1.3 g, 0.8 mL, 11 mmol) was added slowly and dropwise to a methanol (4.3 mL) solution of DL-cyclopropylglycine (0.50 g, 4.3 mmol) that was cooled to 0°C. The reaction mixture was stirred for 10 minutes and then gradually warmed to room temperature. The reaction mixture was stirred at 0°C for 10 min and then gradually warmed to room temperature. Stirring was continued at room temperature overnight. Upon completion of the reaction, the mixture was concentrated and dried under pressure of 0.5 mmHg at room temperature to afford methyl 2-amino-2-cyclopropylacetate hydrochloride (0.60 g, 85% yield) as a white solid. The product was characterized by 1H NMR (DMSO-d6): δ 0.51-0.69, 1.05-1.17, 3.36-3.39, 3.75, 8.69.

10294-18-5
20 suppliers
inquiry

535936-86-8
73 suppliers
$32.00/100mg
Yield: 0.60 g (85%)
Reaction Conditions:
with thionyl chloride in methanol
Steps:
141 methyl amino(cyclopropyl)acetate hydrochloride
Preparation 141 methyl amino(cyclopropyl)acetate hydrochloride To a solution of cyclopropyl glycine (0.50 g, 4.3 mmol) in ice cold MeOH (4.3 mL) was added dropwise SOCl2 (1.3 g, 0.8 mL, 11 mmol). The ensuing mixture was stirred at 0° C. for 10 min then allowed to warm to rt. Stir at rt overnight, concentrate and dry at rt/0.5 mmHg to provide 0.60 g (85%) of methyl amino(cyclopropyl)acetate hydrochloride as a solid: 1H NMR (DMSO-d6) δ0.51-0.69, 1.05-1.17, 3.36-3.39, 3.75, 8.69.
References:
Verhoest, Patrick R.;Hoffman, Robert Louis;Corbett, Jeffrey W.;Ennis, Michael Dalton;Frank, Kristine E.;Fu, Jian-Min US2003/144297, 2003, A1