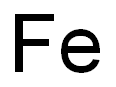methyl 2-amino-4-methanesulfonylbenzoate synthesis
- Product Name:methyl 2-amino-4-methanesulfonylbenzoate
- CAS Number:158580-87-1
- Molecular formula:C9H11NO4S
- Molecular Weight:229.25
Yield:-
Reaction Conditions:
with hydrogenchloride in methanol;dichloromethane;
Steps:
R.16 Reference Example 16
Reference Example 16 Concentrated hydrochloric acid (90 ml) was added to a stirred suspension of methyl 2-nitro-4-methylsulphonyl benzoate (25 g) in methanol at -5° C. (ice/salt bath). The cooling bath was then removed and iron dust (17.5 g) was added portionwise over a period of 20 minutes. The resulting exotherm was controlled using a cooling bath so that the temperature did not exceed 50° C. After 15 minutes of cooling, the bath was removed and the reaction mixture was allowed to reach room temperature. Stirring was continued for a further 3 hours. The mixture was poured onto ice and then neutralised with sodium carbonate. Dichloromethane was added and the suspension was filtered. The filtrate was extracted with further dichloromethane and the combined organic extracts were dried (magnesium sulphate), filtered and evaporated to yield a crude product which was purified by recrystallisation from ethyl acetate/hexane to give methyl 2-amino-4-methylsulphonyl benzoate (4.5 g) as yellow needles, m.p. 98.3-98.5° C. By proceeding in a similar manner, the following compounds were prepared from the appropriately substituted starting material: methyl 2-amino-4-bromobenzoate NMR (CDCl3) 3.8(s,3H), 5.72(bs,2H), 6.7(dd,1H), 6.79(d,1H), 7.65(d,1H);
References:
US6323155,2001,B1