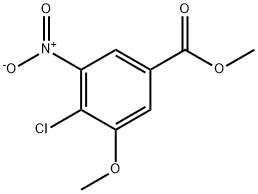
Methyl 4-chloro-3-hydroxy-5-nitrobenzoate synthesis
- Product Name:Methyl 4-chloro-3-hydroxy-5-nitrobenzoate
- CAS Number:180031-12-3
- Molecular formula:C8H6ClNO5
- Molecular Weight:231.59
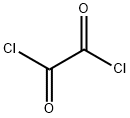
63603-09-8
112 suppliers
$5.00/100mg

180031-12-3
23 suppliers
inquiry
Yield:180031-12-3 94%
Reaction Conditions:
Stage #1: methyl 4-chloro-3-methoxy-5-nitrobenzoatewith boron tribromide in dichloromethane at 20;Cooling with ice;
Stage #2: with sulfuric acid in methanol at 75;
Steps:
6 6. Synthesis of the intermediate compound 4-chloro-3-hydroxy-5-nitrobenzoic acid methyl ester
Disperse 4-chloro-3-methoxy-5-nitrobenzoic acid methyl ester (10g, 40.7mmol) in dry dichloromethane (100mL),Slowly add boron tribromide (40.8g, 162.8mmol) dropwise in an ice bath. After the dropwise addition, it is slowly raised to room temperature and stirred overnight.After the reaction is complete, slowly add methanol dropwise to quench in an ice bath, and then spin dry. Methanol (100 mL) and concentrated sulfuric acid (0.2 mL) were added thereto, and the reaction solution was heated to 75° C. and stirred overnight.After cooling, concentrate under reduced pressure to remove the solvent, add 150 mL of water, and ultrasonically disperse,Filter, wash the solid with water, and dry the solid to obtain4-chloro-3-hydroxy-5-nitrobenzoic acid methyl ester(8.89 g, 38.4 mmol, yield 94%).
References:
CN111471056,2020,A Location in patent:Paragraph 0240-0242