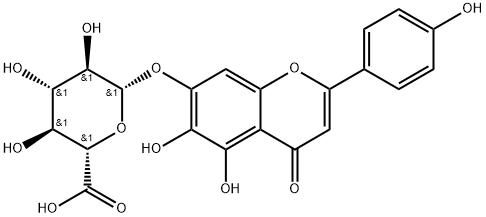
Scutellarin synthesis
- Product Name:Scutellarin
- CAS Number:27740-01-8
- Molecular formula:C21H18O12
- Molecular Weight:462.36
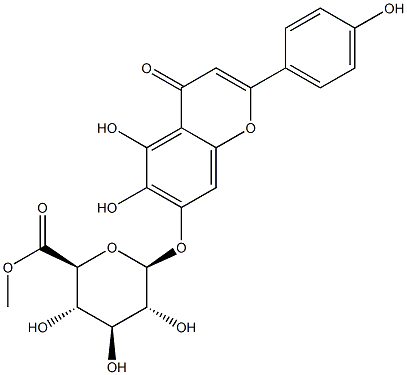
119262-68-9

27740-01-8
General procedure: 20 g (42 mmol) of methyl (2S,3S,4S,5R,6S)-6-((5,6-dihydroxy-2-(4-hydroxyphenyl)-4-yloxy-4H-benzopyran-7-yl)oxy)-3,4,5-trihydroxytetrahydro-2H-pyran-2-carboxylate was dissolved in a mixed solvent of 300 mL of water and 300 mL of acetone at 0 °C Stirring. Under the protection of nitrogen, 70 mL of 10% aqueous sodium hydroxide solution was slowly added dropwise, and the reaction temperature was maintained at 0 °C with continuous stirring for about 2 h. The reaction process was monitored by HPLC. After the reaction was completed, 10% hydrochloric acid aqueous solution was slowly added dropwise under stirring to adjust the pH of the reaction solution to 2.0-3.0, and stirring was continued for 1 hour. Subsequently, the reaction system was gradually brought to room temperature and a large amount of yellow solid 5,6,4'-trihydroxyflavone-7-yloxy-glucuronic acid was observed to precipitate gradually. The reaction mixture was allowed to stand at room temperature overnight, followed by diafiltration and the filter cake was washed with appropriate amount of water and acetone and dried. Finally, purification by ethanol recrystallization afforded 17.3 g of pure 5,6,4'-trihydroxyflavone-7-yloxy-glucuronic acid in 89% yield. The purity of the product was 98.74% by HPLC, and its HPLC chromatogram is shown in Fig. 1, and the statistical results are shown in Table 1.1H-NMR (400 MHz, DMSO) data were as follows: δ 10.41 (1H, s), 8.63 (1H, s), 7.94 (2H, d, J = 8.8Hz), 6.99 (1H, s), 6.94 (2H, d, J = 8.8Hz), 6.99 (1H, s), 6.94 (2H, d, J = 8.8Hz). d, J = 8.8 Hz), 6.86 (1H, s), 5.51-5.31 (3H, br), 5.28 (1H, d, J = 7.5 Hz), 4.11 (2H, d, J = 9.6 Hz), 3.43 (4H, m).

119262-68-9
132 suppliers
inquiry

27740-01-8
272 suppliers
$31.00/5mg
Yield:27740-01-8 89%
Reaction Conditions:
Stage #1:scutellarein 7-O-β-D-glucuronopyranoside methyl ester with water;sodium hydroxide in acetone at 0; for 0.2 h;Inert atmosphere;
Stage #2: with hydrogenchloride in water;acetone; pH=2-3 for 1 h;Reagent/catalyst;Temperature;Time;Concentration;
Steps:
1.4 4. Preparation of 5,6,4'-trihydroxyflavone-7-O-D-glucuronic acid
A mixed solution of 300 mL water and 300 mL acetone was added to 20 g (42 mmol) of compound IV, and 70 mL of 10% aqueous sodium hydroxide was added dropwise under stirring at 0 °C. The reaction was stirred continuously under protection of nitrogen gas at 0 °C for about two hours, which was completed as detected by HPLC. 10% aqueous hydrochloric acid was added dropwise under stirring, to adjust pH to 2.0∼3.0, and stirred continuously for 1 hour followed by gradually raising to room temperature, with large amount of yellow solid 5,6,4'-trihydroxyflavone-7-O-D-glucuronic acid gradually separated out. The reaction mixture was left standing overnight at room temperature, filtered by suction, and the filter cake was washed with suitable amount of water and acetone, dried and then recrystallized with ethanol to obtain 17.3 g of pure 5,6,4'-trihydroxyflavone-7-O-D-glucuronic acid, with a yield of 89%. The purity was 98.74% detected by HPLC, the chromatogram detected by HPLC was shown in Figure 1, and the statistical result was shown in table 1. 1H-NMR (400MHz, DMSO), δ (ppm): 10.41 (1H, s), 8.63 (1H, s), 7.94 (2H, d, J= 8.8Hz), 6.99 (1H, s), 6.94 (2H, d, J= 8.8Hz), 6.86 (1H, s), 5.51-5.31 (3H, br), 5.28 (1H, d, J= 7.5 Hz), 4.11 (2H, d, J=9.6Hz), 3.43 (4H, m).
References:
Kunming Pharmaceutical Corp.;LI, Penghui;ZHANG, Wei;YANG, Zhaoxiang;ZHANG, Xiongbo;WANG, Jun;ZHU, Haibei;CHEN, Jinxin;BAI, Yiyang EP2840088, 2015, A1 Location in patent:Paragraph 0042
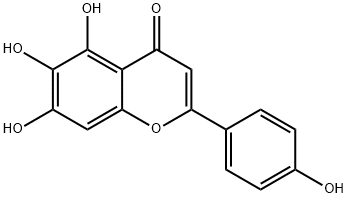
529-53-3
238 suppliers
$25.00/2mg

27740-01-8
272 suppliers
$31.00/5mg
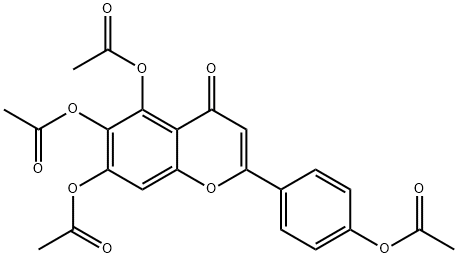
1180-46-7
11 suppliers
inquiry

27740-01-8
272 suppliers
$31.00/5mg

529-53-3
238 suppliers
$25.00/2mg
![(2S,3S,4R,5R,6R)-6-[[[(2S,3S,4R,5R)-5-(2,4-dioxopyrimidin-1-yl)-3,4-dihydroxy-oxolan-2-yl]methoxy-hydroxy-phosphoryl]oxy-hydroxy-phosphoryl]oxy-3,4,5-trihydroxy-oxane-2-carboxylic](/CAS/20210111/GIF/2616-64-0.gif)
2616-64-0
73 suppliers
inquiry

27740-01-8
272 suppliers
$31.00/5mg