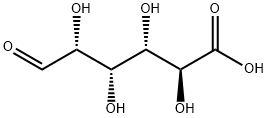
D-Glucuronic acid
- CAS No.
- 6556-12-3
- Chemical Name:
- D-Glucuronic acid
- Synonyms
- GLUCURONIC ACID;glucuronate;D-glucuronate;(2S,3S,4S,5R)-2,3,4,5-Tetrahydroxy-6-oxohexanoic acid;D-GLCA;Glucosiduronic Acid;Glucuronic Aci;D-GlucuronicAci;D-Glucurono acid;Glycuronic acid
- CBNumber:
- CB9303344
- Molecular Formula:
- C6H10O7
- Molecular Weight:
- 194.14
- MDL Number:
- MFCD00077778
- MOL File:
- 6556-12-3.mol
- MSDS File:
- SDS
| Melting point | 159-161 °C (lit.) |
|---|---|
| alpha | [α]D20 +35.0~+39.0゜(c=6,H2O) |
| Boiling point | 250.56°C (rough estimate) |
| Density | 1.4301 (rough estimate) |
| refractive index | 36 ° (C=6, H2O) |
| storage temp. | -20°C |
| solubility | H2O: soluble100mg/mL, clear to slightly hazy |
| pka | pKa 3.18(H2O t=20.0) (Uncertain) |
| form | Crystalline Powder |
| color | White to off-white |
| Water Solubility | Soluble in water. |
| Merck | 14,4465 |
| BRN | 1727083 |
| Stability | Hygroscopic |
| InChIKey | LCUVNMXNGSEBHT-RJOKPDHXSA-N |
| CAS DataBase Reference | 6556-12-3(CAS DataBase Reference) |
| EWG's Food Scores | 1 |
| NCI Dictionary of Cancer Terms | glucuronic acid |
| FDA UNII | 8A5D83Q4RW |
| NCI Drug Dictionary | glucuronic acid |
| EPA Substance Registry System | D-Glucuronic acid (6556-12-3) |
SAFETY
Risk and Safety Statements
| Symbol(GHS) |  GHS07 |
|||||||||
|---|---|---|---|---|---|---|---|---|---|---|
| Signal word | Warning | |||||||||
| Hazard statements | H315-H319-H335 | |||||||||
| Precautionary statements | P261-P305+P351+P338 | |||||||||
| Hazard Codes | Xi | |||||||||
| Risk Statements | 36/37/38 | |||||||||
| Safety Statements | 37/39-26-36 | |||||||||
| WGK Germany | 3 | |||||||||
| RTECS | LZ8836600 | |||||||||
| TSCA | Yes | |||||||||
| HS Code | 2932 99 00 | |||||||||
| NFPA 704 |
|
D-Glucuronic acid price More Price(42)
| Manufacturer | Product number | Product description | CAS number | Packaging | Price | Updated | Buy |
|---|---|---|---|---|---|---|---|
| Sigma-Aldrich | G5269 | D-Glucuronic acid ≥98% (GC) | 6556-12-3 | 10mg | $35 | 2024-03-01 | Buy |
| Sigma-Aldrich | 8.43961 | D-Glucoronic acid for synthese | 6556-12-3 | 10G | $128 | 2024-03-01 | Buy |
| Sigma-Aldrich | 8.43961 | D-Glucoronic acid for synthese | 6556-12-3 | 25G | $254 | 2024-03-01 | Buy |
| Sigma-Aldrich | 1294319 | D-Glucuronic acid United States Pharmacopeia (USP) Reference Standard | 6556-12-3 | 50mg | $519 | 2024-03-01 | Buy |
| TCI Chemical | G0302 | D-Glucuronic Acid >96.0%(HPLC)(T) | 6556-12-3 | 5g | $38 | 2024-03-01 | Buy |
D-Glucuronic acid Chemical Properties,Uses,Production
Description
Glucuronic acid (from Ancient Greek γλυκ?? "sweet" + ο?ρον "urine") is a carboxylic acid. Its structure is similar to that of glucose. However, glucuronic acid's sixth carbon is oxidized to a carboxylic acid. Its formula is C6H10O7. The salts and esters of glucuronic acid are known as glucuronates; the anion C6H9O7-is the glucuronate ion.
Chemical Properties
White Solid
Uses
D-Glucuronic acid is used as pharmaceutical intermediate and in chemical research.
Uses
- D-Glucuronic acid is widely distributed in the plant and animal kingdoms. D-Glucuronic acid usually occurs in "paired" form as a glycosidic combination with phenols, alcohols. Such glucuronides form in the liver to detoxify poisonous hydroxyl-containing substances. The glucuronides present in normal urine are those of phenol, cresol, and indoxyl. After the ingestion of poisons such as morphine, chloral hydrate, camphor, or turpentine, glucuronides formed with the poison or its hydroxylated derivatives appear in the urine.
- Chiral D-Glucuronic acid is a basic building block of hyaluronic acid and chondroitin sulfate. It is a precursor to vitamin C, the chief detoxifying agent in plants and animals.
- D-Glucuronic Acid is a sugar acid formed by the oxidation of the C-6 carbon of GLUCOSE. In addition to being a key intermediate metabolite of the uronic acid pathway, glucuronic acid also plays a role in the detoxification of certain drugs and toxins by conjugating with them to form GLUCURONIDES.
Uses
D-glucuronic acid and D-galacturonic acid are naturally occurring hexuronic acids present in glycosaminoglucans, glucuronide conjugates in mammals and in plant cell wall polysaccharides.
D-Glucuronic acid exists as a component of glycosaminoglucans such as hyaluronan, heparin and chondroitin sulphate present in mammalian connective tissue such as cartilage. In the synthesis of glucuronide conjugates in mammals glucuronic acid is conjugated to xenobiotic compounds, a biosynthesis reaction known as glucuronidation, catalysed by the enzyme UDP-glucuronyltransferase and considered a detoxifying process.
Both D-glucuronic acid and D-galacturonic acid are major components of plant cell wall polysaccharides. D-glucuronic acid is a component of arabinoxylan, which consists of a β-(1,4)-linked xylan backbone substituted with α-(1-2/3)-linked L-arabinofuranose and α-(1-2)- linked 4-O-methylglucuronic acid. D-Galacturonic acid is the major component of pectin comprising the α-(1,4)-linked galacturonan backbone of homogalacturonan and rhamnogalacturonan II, and is present within the repeating disaccharide unit [,4)-α-D-GalpA-(1,2)-α- L-Rhap-(1,] of rhamnogalacturonan I.
Definition
ChEBI: A D-glucopyranuronic acid in which the anomeric centre has alpha-configuration.
Definition
glucuronic acid: A compound,OC6H9O6, derived from the oxidationof glucose. It is an important constituentof gums and mucilages. Glucuronicacid can combine withhydroxyl (–OH), carboxyl (–COOH), oramino (-NH2) groups to form a glucuronide.The addition of a glucuronidegroup to a molecule(glucuronidation) generally increasesthe solubility of a compound; henceglucuronidation plays an importantrole in the excretion of foreign substances.
Biological Functions
Proteoglycans Glucuronic acid is common in carbohydrate chains of proteoglycans. It is part of mucous animal secretions (such as saliva), cell glycocalyx and intercellular matrix (for instance hyaluronan))
Glucuronidation of toxic substances
In the animal body, glucuronic acid is often linked to the xenobiotic metabolism of substances such as drugs, pollutants, bilirubin, androgens, estrogens, mineralocorticoids, glucocorticoids, fatty acid derivatives, retinoids, and bile acids. These linkages involve glycosidic bonds, and this linkage process is known as glucuronidation. Glucuronidation occurs mainly in the liver, although the enzyme responsible for its catalysis, UDP-glucuronyltransferase, has been found in all major body organs, e.g., intestine, kidneys, brain, adrenal gland, spleen, and thymus.
Use
Determination of urinary steroids and of steroid conjugates in blood. Contained in some commercially available brands of Kombucha as an antioxidant & organic acid In all plants and mammals - other than guinea pigs and primatesglucuronic acid is a precursor of ascorbic acid , also known as vitamin c.
ConformationUnlike its C5 epimer iduronic acid, which may occur in a number of conformations, glucuronic acid occurs in predominantly the 4C1 conformation.
Glucuronidases
Glucuronidases are those enzymes that hydrolyze the glycosidic bond between glucuronic acid and some other compound.
General Description
Pharmaceutical secondary standards for application in quality control provide pharma laboratories and manufacturers with a convenient and cost-effective alternative to the preparation of in-house working standards
Biochem/physiol Actions
In humans, glucuronic acid conjugation with steroidal compounds and drugs is an important step in phase II metabolic reactions. This influences the biotransformation process of the compound.
Purification Methods
Crystallise the acid from EtOH or EtOAc, wash it with MeOH and dry it in vacuo to give the “ ” form. Heating converts it to the lactone (see below). The sodium salt monohydrate [207300-70-7] M 234.1 has m ~136-138o(dec) [] D 20 +21o (c 2, H2O after 2hours). [Sutter & Reichstein Helv Chim Acta 21 1210 1938, Beilstein 3 H 886, 3 IV 1997.]
D-Glucuronic acid Preparation Products And Raw materials
Raw materials
Preparation Products
| Supplier | Tel | Country | ProdList | Advantage | |
|---|---|---|---|---|---|
| Anhui Hegeng Biotech Engineering Co., Ltd. | 21-65953622 +8613916053686 | sales@hegengbt.com | China | 10 | 58 |
| Shanghai Daken Advanced Materials Co.,Ltd | +86-371-66670886 | info@dakenam.com | China | 15928 | 58 |
| Henan Tianfu Chemical Co.,Ltd. | +86-0371-55170693 +86-19937530512 | info@tianfuchem.com | China | 21691 | 55 |
| Hangzhou FandaChem Co.,Ltd. | 008657128800458; +8615858145714 | fandachem@gmail.com | China | 9348 | 55 |
| Xi'an Kono chem co., Ltd., | 029-86107037 13289246953 | info@konochemical.com | China | 2995 | 58 |
| Hubei xin bonus chemical co. LTD | 86-13657291602 | linda@hubeijusheng.com | CHINA | 22968 | 58 |
| BOC Sciences | +1-631-485-4226 | inquiry@bocsci.com | United States | 19553 | 58 |
| Shanghai Standard Technology Co., Ltd. | 18502101150 | ft-sales@nature-standard.com | CHINA | 1923 | 58 |
| CONIER CHEM AND PHARMA LIMITED | +8618523575427 | sales@conier.com | China | 49390 | 58 |
| career henan chemical co | +86-0371-86658258 15093356674; | factory@coreychem.com | China | 29826 | 58 |
View Lastest Price from D-Glucuronic acid manufacturers
| Image | Update time | Product | Price | Min. Order | Purity | Supply Ability | Manufacturer | |
|---|---|---|---|---|---|---|---|---|
 |
2023-02-24 | D-Glucuronic acid
6556-12-3
|
US $0.00 / mg | 20mg | ≥98%(HPLC) | 10 g | Shanghai Standard Technology Co., Ltd. | |
 |
2021-09-29 | D-Glucuronic acid
6556-12-3
|
US $0.00 / KG | 1KG | 99% | 500kgs/month | WUHAN FORTUNA CHEMICAL CO., LTD | |
 |
2021-07-13 | D-GLUCURONIC ACID
6556-12-3
|
US $15.00-10.00 / KG | 1KG | 99%+ HPLC | Monthly supply of 1 ton | Zhuozhou Wenxi import and Export Co., Ltd |
-
- D-Glucuronic acid
6556-12-3
- US $0.00 / mg
- ≥98%(HPLC)
- Shanghai Standard Technology Co., Ltd.
-

- D-Glucuronic acid
6556-12-3
- US $0.00 / KG
- 99%
- WUHAN FORTUNA CHEMICAL CO., LTD
-

- D-GLUCURONIC ACID
6556-12-3
- US $15.00-10.00 / KG
- 99%+ HPLC
- Zhuozhou Wenxi import and Export Co., Ltd
6556-12-3(D-Glucuronic acid)Related Search:
1of4