クロルプロマジン 化学特性,用途語,生産方法
解説
2-chloro-10-[(3-dimethylamino)propyl]phenothiazine.C17H19ClN2S(318.87).2-クロロフェノチアジンにナトリウムアミドの存在下で3-(ジメチルアミノ)プロピルクロリドを作用させると得られる.
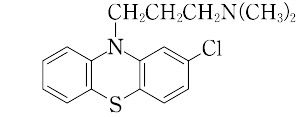
"アミン臭のある油状液体.沸点200~205 ℃(0.1 kPa).精神分裂症に用いられるトランキライザー.少量で鎮静作用を,大量で睡眠を起こさせる.塩酸塩は白色の結晶.融点194~196 ℃.LD50 405 mg/kg(マウス,経口)[CAS 50-53-3][CAS 14923-91-2:塩酸塩]
森北出版「化学辞典(第2版)
用途
抗精神病薬統合失調症,非定型精神病,中毒性精神病,器質性精神病などに用いられ,躁うつ病にも補助的に用いられる 。
効能
統合失調症治療薬, 制吐薬
説明
This phenothiazine with sedative properties is used in
human medicines and has induced contact dermatitis
in nurses or in those working in the pharmaceutical
industry. It is also used in veterinary medicine to avoid
mortality of pigs during transportation. It is a sensitizer
and a photosensitizer.
化学的特性
Oily liquid; amine odor.
使用
In psychiatric practice, chlorpromazine is used in various conditions of psychomotor
excitement in patients with schizophrenia, chronic paranoid and also manic-depressive
conditions, neurosis, alcohol psychosis and neurosis accompanied by excitement, fear,
stress, and insomnia. In comparison with other neuroleptics, chlorpromazine is unique in
that it has an expressed sedative effect. It is sometimes used in anesthesiological practice
for potentiating narcosis. It also has moderate anticonvulsant action.
定義
ChEBI: A substituted phenothiazine in which the ring nitrogen at position 10 is attached to C-3 of an N,N-dimethylpropanamine moiety.
危険性
Toxic by ingestion.
接触アレルゲン
This phenothiazine with sedative properties is used in
human medicine and induced contact dermatitis in nurses
or those working in the pharmaceutical industry. It is also
used in veterinary medicine to avoid mortality of pigs during
transportation. It is a sensitizer and a photosensitizer.
環境運命予測
Acute and chronic toxicity due to chlorpromazine generally
manifests as an extension of normal pharmacological activity.
The precise mechanism of action of chlorpromazine, and other
phenothiazines, is unknown; however, it is thought to primarily
involve antagonism of dopaminergic (D2) neurotransmission
at synaptic sites and blockade of postsynaptic dopamine
receptor sites at the subcortical levels of the reticular formation,
limbic system, and hypothalamus. This activity contributes to
chlorpromazine’s extrapyramidal reactions. Chlorpromazine
also has strong central and peripheral activity directed against
adrenergic receptors and weak activity against serotonergic,
histaminic (H1), and muscarinic receptors. Chlorpromazine
has slight ganglionic blocking action. Chlorpromazine is
known to depress vasomotor reflexes medicated by the hypothalamus
and/or brain stem; inhibit release of growth hormone;
antagonize secretion of prolactin release-inhibiting hormone;
and reduce secretion of corticotropin-regulatory hormone.
Chlorpromazine also has direct effects on cardiac myocytes;
it can induce early after-depolarizations, block depolarizing
sodium channels, and cause significant prolongation of the
QTc interval.
Chlorpromazine may be irritating to eyes, mucous
membranes, and skin. Contact and inhalation should be
avoided.
代謝経路
The in vivo photodegradation of chlorpromazine in rat
skin exposed to UV-A results in the formation of
promazine and 2-hydroxypromazine in irradiated rats,
but not in the skin of rats kept in the dark.
Chlorpromazine sulfoxide is a major metabolite of
chlorpromazine, found in smaller quantity in the skin of
irradiated rats compared with those kept in the dark.
Chlorpromazine sulfoxide is not a photoproduct of
chlorpromazine under the experimental conditions.
クロルプロマジン 上流と下流の製品情報
原材料
準備製品