- U-69593
-

- $1.00 / 1g
-
2024-04-29
- CAS:96744-75-1
- Min. Order: 1g
- Purity: 99%
- Supply Ability: 100kg
- U-69593
-

- $0.00 / 1kg
-
2024-04-12
- CAS:96744-75-1
- Min. Order: 1kg
- Purity: 0.99
- Supply Ability: 10T
- U-69593
-
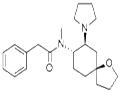
- $1.00 / 1KG
-
2019-07-06
- CAS:96744-75-1
- Min. Order: 1G
- Purity: 98%
- Supply Ability: 100KG
|
| | U-69593 Basic information |
| Product Name: | U-69593 | | Synonyms: | U-69593;(+)-(5ALPHA,7ALPHA,8BETA)-N-METHYL-N-[7-(1-PYRROLIDINYL)-1-OXASPIRO[4.5]DEC-8-YL]-BENZENEACETAMIDE;(5ALPHA,7ALPHA,8BETA)-(+)-N-METHYL-N-(7-[1-PYRROLIDINYL]-1-OXASPIRO[4.5]DEC-8-YL)-BENZENEACETAMIDE;U-69593 SELECTIVE K OPIOID AG;(5A,7A,8B)-(-)-N-METHYL-(7-(1-PYRROLIDINYL)-1-OXASPIRO (4.5)DEC-8- YL)BENZENEACETAMIDE);Benzeneacetamide, N-methyl-N-((5R,7S,8S)-7-(1-pyrrolidinyl)-1-oxaspiro(4.5)dec-8-yl)-;Benzeneacetamide, N-methyl-N-(7-(1-pyrrolidinyl)-1-oxaspiro(4.5)dec-8-yl)-, (5alpha,7alpha,8beta)-(-)-;N-Methyl-N-(7-(1-pyrrolidinyl)-1-oxaspiro(4,5)dec-8-yl)benzeneacetamide | | CAS: | 96744-75-1 | | MF: | C22H32N2O2 | | MW: | 356.5 | | EINECS: | | | Product Categories: | | | Mol File: | 96744-75-1.mol | 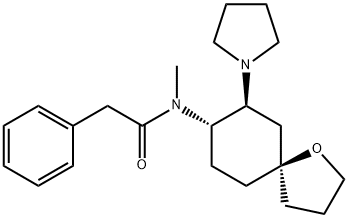 |
| | U-69593 Chemical Properties |
| Melting point | 120-123℃ | | Boiling point | 518.7±50.0 °C(Predicted) | | density | 1.13 | | RTECS | CY1490060 | | storage temp. | 2-8°C | | solubility | H2O: 14 mg/mL | | form | solid | | pka | 9.15±0.20(Predicted) | | color | white | | optical activity | [α]/D +7.8°, c = 0.825 in methanol(lit.) |
| | U-69593 Usage And Synthesis |
| Uses | U-69593 is a selective κ-opioid receptor agonist. A safe opioid analgesic. | | Definition | ChEBI: U69593 is a monocarboxylic acid amide obtained by formal condensation between the carboxy group of phenylacetic acid and the secodary amino group of (5R,7S,8S)-N-methyl-7-(pyrrolidin-1-yl)-1-oxaspiro[4.5]decan-8-amine. It has a role as a kappa-opioid receptor agonist, an anti-inflammatory agent and a diuretic. It is an oxaspiro compound, a N-alkylpyrrolidine, an organic heterobicyclic compound and a monocarboxylic acid amide. | | Biological Functions | U-69593 is a single enantiomer with very high selectivity for the kappa receptor in vitro (mu/kappa ratio = 484). It was radiolabelled by a catalytic exchange of tritium for the two aromatic chlorine substituents of the precursor. This ligand is commercially available and has been widely used as a radiolabel for the kappa receptor.
in spinal cordslice preparations from the 9-16-day-old rat,U-69593 produced a na-loxone-reversible depression of spontaneous and electrically evoked activityin dorsal horn neurones. | | Biochem/physiol Actions | U-69593 is a selective κ opioid receptor agonist. U-69593 is known to inhibit cocaine sensitization in meso-limbic dopamine neurons by normalizing basal overflow of dopamine. | | Structure and conformation | An X-ray analysis of the crystal structures of U-50488 and U-69593 have been determined. The cyclohexane ring adopts a chair conformation with both nitrogen substituents attached equatorially The amide bond is in a trans conformation and the absolute stereochemistry of the kappa opioid active enantiomer of U-50488 is [S,S]. There appears to be some confusion concerning the ab-solute stereochemistry of U-69593.The structure in the publication describ-ing the X-ray crystallography is drawn as 5R,7S,8S,but the title of thispaper,which gives the full chemical name of U-69593, indicates 5S,7S,8S. |
| | U-69593 Preparation Products And Raw materials |
|