| Company Name: |
BOC Sciences
|
| Tel: |
1-631-485-4226; 16314854226 |
| Email: |
info@bocsci.com |
| Products Intro: |
Product Name:Pactamycin
CAS:23668-11-3
Purity:>=95% Remarks:It is produced by the strain of Str. pactum var. pactum NRRL 2939. It is a cyclopentane and cyclohex
|
|
| | Pactamycin Basic information |
| Product Name: | Pactamycin | | Synonyms: | A 80856F30;NSC52947;Pactamycin;2-Hydroxy-6-methylbenzoic acid [(1S)-5α-[(3-acetylphenyl)amino]-4β-amino-3β-[[(dimethylamino)carbonyl]amino]-1β,2α-dihydroxy-3-[(S)-1-hydroxyethyl]-2-methylcyclopentyl]methyl ester;2-Hydroxy-6-methylbenzoic acid [[(1S)-5α-(3-acetylphenylamino)-4β-amino-3β-(3,3-dimethylureido)-1β,2α-dihydroxy-3-[(S)-1-hydroxyethyl]-2-methylcyclopentan-1α-yl]methyl] ester;2-Hydroxy-6-methylbenzoic acid [(1S,2R,3R,4S,5S)-5-[(3-acetylphenyl)amino]-4-amino-3-[[(dimethylamino)carbonyl]amino]-1,2-dihydroxy-3-[(1S)-1-hydroxyethyl]-2-methylcyclopentyl]methyl ester;PNU-0015800;U-15800 | | CAS: | 23668-11-3 | | MF: | C28H38N4O8 | | MW: | 558.62 | | EINECS: | | | Product Categories: | | | Mol File: | 23668-11-3.mol | 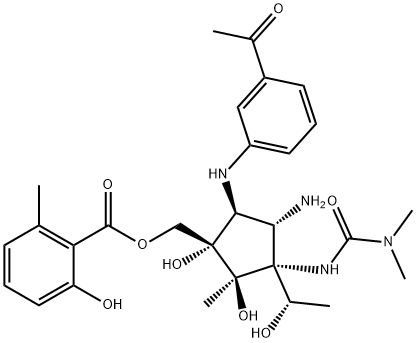 |
| | Pactamycin Chemical Properties |
| Boiling point | 626.7°C (rough estimate) | | alpha | D25 +79° (ethanol) changing to +23° on standing; changes in acetone on standing from +25° to +76° | | density | 1.2608 (rough estimate) | | refractive index | 1.5500 (estimate) | | storage temp. | 2-8°C | | solubility | DMSO: ≥12mg/mL | | pka | 8.10±0.35(Predicted) | | color | white to tan |
| Hazard Codes | T+ | | Risk Statements | 28-36/37/38 | | Safety Statements | 26-28-36/37-45 | | RIDADR | UN 2811 6.1 / PGII | | Toxicity | LD50 in mice (mg/kg): 10.7 orally; 15.6 i.v.; in rats (mg/kg): 1.4 i.v. (Bhuyan) |
| | Pactamycin Usage And Synthesis |
| Safety Profile | Poison by ingestion, intravenous, and intraperitoneal routes. Mutation data reported. When heated to decomposition it emits toxic fumes of NOx. | | Enzyme inhibitor | This universal translation inhibitor (FW = 558.63 g/mol; CAS 23668-11-3; soluble in ethanol and benzene; Abbreviation: Pct) from Streptomyces pactum, previously known as NSC 52947 and named systematically as benzoic acid, 2-hydroxy-6-methyl-[(1S,2R,3R,4S,5S)-5-[(3-acetylphenyl) amino]-4-amino-3-[[(dimethylamino)carbonyl]amino]-1,2-dihydroxy-3- [(1S)-1-hydroxyethyl]-2-methylcyclopentyl]methyl ester, inhibits protein synthesis and exhibits antitumor activity. Because the optical rotation of dissolved pactamycin changes upon standing, some as-yet undefined chemical rearrangement must take place. Solutions should be freshly prepared. Primary Mode of Inhibitory Action: Pct restricts structural transitions in ribosomal RNA, preventing the ribosome blocks the binding of initiator tRNA to the initiator complex, preventing formation of the 80S ribosomal complex and arresting protein biosynthesis immediately after the initial dipeptide is formed. Binding Interactions: Pct binds in a single site on the 30S ribosome subunit in the upper part of the platform, very close to the cleft in the subunit that is responsible for binding of the three tRNA molecules. It interacts primarily with residues at the tips of the stem loops H23b and H24a in the central domain of 16S RNA, where it folds up to mimic an RNA dinucleotide. The interaction between N6 of A694 and Pct is crucial for binding of the drug since this is the only interaction with that particular base. N-methylation of this residue causes resistance, most likely from distortions of the local structure |
| | Pactamycin Preparation Products And Raw materials |
|