|
|
| | Benzyl vinylcarbamate Basic information |
| Product Name: | Benzyl vinylcarbamate | | Synonyms: | O-Benzyl-N-vinylcarbamate;N-Vinyl-O-benzyl urethane;BENZYL-N-VINYLCARBAMATE;Phenylmethyl ethenylcarbamate;benzyl N-ethenylcarbamate;Carbamic acid, N-ethenyl-, phenylmethyl ester;benzyl ethenylcarbamate;benzyl vinylcarbamate | | CAS: | 84713-20-2 | | MF: | C10H11NO2 | | MW: | 177.2 | | EINECS: | | | Product Categories: | organic building block | | Mol File: | 84713-20-2.mol | 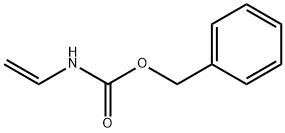 |
| | Benzyl vinylcarbamate Chemical Properties |
| Melting point | 43-44°C | | Boiling point | 272℃ | | density | 1.089 | | Fp | 118℃ | | storage temp. | Keep in dark place,Inert atmosphere,Store in freezer, under -20°C | | solubility | Chloroform (Slightly), Ethyl Acetate (Slightly) | | form | Solid | | pka | 11.04±0.46(Predicted) | | color | White to Off-White |
| | Benzyl vinylcarbamate Usage And Synthesis |
| Uses | Benzyl Vinylcarbamate is used as a reagent in the synthesis of CF3- or -?CF2-?substituted tetrahydroquinolines. | | Preparation | A 1-L flask was equipped with a variable speed pump, a mechanical stirrer, a temperature controller, a 4" (10 cm) column packed with ceramic saddles, a distillation head, a spiral condenser (cooled with water at 10–15 ℃), and a receiver. The flask was charged with toluene (150–200 mL) and phenothiazine (0.5 g) and the solution was heated to 105–110 ℃. The receiver was charged with benzyl alcohol (86 g, 0.8 mol), phenothiazine (0.05 g), and triethylamine (0.1–0.3 g). This mixture was cooled in ice and stirred. A solution of acryloyl azide (1 mol), prepared as described above, was pumped into the distillation flask over a period of 4–5 h, maintaining the pot temperature at 105–110 ℃ with a heating mantle. The vapor temperature varied, depending on the rate of addition of the azide, but was in the range 80–100 ℃. The distillate was passed directly into the benzyl alcohol mixture. After the addition of acryloyl azide, the distillation continued, generating a further 10–20 mL of toluene. The receiver was then removed from the distillation set-up, and its contents were stirred at 0–5 ℃ for 1– 2 h. The product mixture was then allowed to gradually warm to room temperature and was stirred until HPLC analysis indicated complete reaction. The mixture was then concentrated in vacuo to a weight of 200–250 g. The residue was treated with heptane (300–350 mL) and cooled to 15 ℃ with stirring. A few seed crystals of benzyl N-vinyl carbamate 810 were added, and the mixture was stirred for 2–3 h. The product was collected by filtration, washed with heptane, and dried in vacuo. Yield 115–128 g (65–72%); mp 41–44 ℃. | | Application |
Benzyl-N-vinyl carbamate (Z-vinylamine; Benzyl vinylcarbamate) is a valuable synthetic intermediate. This compound undergoes alkylation readily on the carbon R to the nitrogen, a property that has been used in the synthesis of a-lactam antibiotics. Z-vinylamine can be readily polymerized into polyvinyl amine derivatives[1].
| | Synthesis Reference(s) | The Journal of Organic Chemistry, 54, p. 4649, 1989 DOI: 10.1021/jo00280a036 | | References | [1] Govindan, Cheruthur K. “An Improved Process for the Preparation of Benzyl-N-vinyl Carbamate1.” Organic Process Research Development 6 1 (2001): 74–77.
|
| | Benzyl vinylcarbamate Preparation Products And Raw materials |
|