| Company Name: |
GIHI CHEMICALS CO.,LIMITED |
| Tel: |
+8618058761490 |
| Email: |
info@gihichemicals.com |
| Products Intro: |
Product Name:2,5-Epoxycyclopenta[5,6]naphth[1,2-d]azepin-9-ol,octadecahydro-5a,7a-dimethyl-, (2S,5R,5aS,5bS,7aR,9S,10aS,10bS,12aR)-
CAS:467-51-6
Purity:99 Package:5KG;1KG,25kg
|
|
|
|
|
|
| | SAMANDARINE Basic information |
| Product Name: | SAMANDARINE | | Synonyms: | SAMANDARINE;1α,4α-Epoxy-3-aza-A-homo-5β-androstan-16β-ol;Samandarin;2,5-Epoxycyclopenta[5,6]naphth[1,2-d]azepin-9-ol, octadecahydro-5a,7a-dimethyl-, (2S,5R,5aS,5bS,7aR,9S,10aS,10bS,12aR)- | | CAS: | 467-51-6 | | MF: | C19H31NO2 | | MW: | 305.45 | | EINECS: | | | Product Categories: | | | Mol File: | 467-51-6.mol | 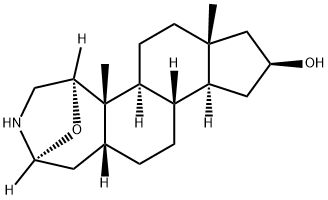 |
| | SAMANDARINE Chemical Properties |
| Melting point | 187-188° | | alpha | D17 +43.3° (acetone) | | Boiling point | 427.9±40.0 °C(Predicted) | | density | 1.111±0.06 g/cm3(Predicted) | | pka | 15.26±0.60(Predicted) | | Optical Rotation | +43.317 (c 0.842, acetone) |
| | SAMANDARINE Usage And Synthesis |
| Description | This pentacyclic alkaloid also occurs in the skin secretion of Salamandra species, particularly S. atra and S. maculosa. It forms colourless crystals from a variety of solvents but when crystallized from MeOH contains solvent of crystallization. The solutions are strongly alkaline and the base is dextrorotatory with [α]17D + 43.7°. Two bridge methyl groups, a non-phenolic hydroxyl group and a methylimino group are present in the molecule. Several crystalline derivatives have been prepared including the hydrochloride, m.p. 32l-2°C; the O,N-diacetyl derivative, m.p. l67-8°C; the monoformyl compound, m.p. l48-ISOoC; diformyl derivative, m.p. 2S6-g0C; N-nitroso compound, m.p. 164-5°C and the N-methyl derivative which is characterized as the hydrochloride, m.p. 300-2°C (dec.) and the methiodide, m.p. 27l-2°C. | | Definition | ChEBI: Samandarine is a steroid alkaloid fundamental parent. | | References | Schopf, Braun., Annalen., 514,69 (1934)
Schopf, Koch., ibid, 552,62 (1942)
Wolfeletal., Ber., 94, 2361 (1961) |
| | SAMANDARINE Preparation Products And Raw materials |
|