|
|
| | 9-methoxycamptothecin Basic information |
| Product Name: | 9-methoxycamptothecin | | Synonyms: | 9-methoxycamptothecin;(4S)-9-Methoxy-4α-ethyl-4β-hydroxy-1H-pyrano[3',4':6,7]indolizino[1,2-b]quinoline-3,14(4H,12H)-dione;(4S)-4-Ethyl-4-hydroxy-9-methoxy-1H-pyrano[3',4':6,7]indolizino[1,2-b]quinoline-3,14(4H,12H)-dione;10-MethoxycaMptothecine;1H-Pyrano[3',4':6,7]indolizino[1,2-b]quinoline-3,14(4H,12H)-dione,4-ethyl-4-hydroxy-9-methoxy-, (4S)-;product/154918;10 Methoxycamptothecin,Inhibitor,10Methoxycamptothecin,inhibit,10-Methoxycamptothecin | | CAS: | 19685-10-0 | | MF: | C21H18N2O5 | | MW: | 378.38 | | EINECS: | 2017-001-1 | | Product Categories: | chemical reagent;pharmaceutical intermediate;phytochemical;reference standards from Chinese medicinal herbs (TCM).;standardized herbal extract | | Mol File: | 19685-10-0.mol | 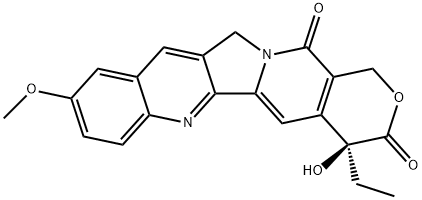 |
| | 9-methoxycamptothecin Chemical Properties |
| Melting point | 254-255℃ | | Boiling point | 773.1±60.0 °C(Predicted) | | density | 1.50 | | storage temp. | 4°C, protect from light | | solubility | Soluble in Chloroform,Dichloromethane,Ethyl Acetate,DMSO,Acetone,etc. | | form | Cryst. | | pka | 11.21±0.20(Predicted) |
| | 9-methoxycamptothecin Usage And Synthesis |
| Description | A minor constituent of Mappia foetida Miers. The alkaloid yields pale yellow
crystals when purified from CHC13-MeOH. It is laevorotatory with [α]24D -
77.5° (c 1.0, pyridine). The structure has been established by spectroscopic
methods. | | Uses | 9-Methoxycamptothecin is an alkaloid derivative of Camptothecin (C175150) derivative used in the treatment of cancers. Camptothecin is a potent anti-cancer agent. | | Uses | 10-Methoxycamptothecin is a Camptothecin (C175150) derivative used in the treatment of cancers. Used in the treatment of hyperproliferative diseases. | | References | Govindachari, Viswanathan., Phytochem., 11, 3529 (1972)
Govindachari, Viswanathan.,Ind. J. Chem., 10,453 (1972) |
| | 9-methoxycamptothecin Preparation Products And Raw materials |
|