|
|
| | 4-(2,4-DINITROANILINO)PHENOL Basic information |
| Product Name: | 4-(2,4-DINITROANILINO)PHENOL | | Synonyms: | acetoquinonelightyellow2rz;Amacel Yellow RR;amacelyellowrr;C.I. 10345;C.I. Disperse Yellow 1;C.I. Solvent yellow 52;c.i.10345;c.i.disperseyellow1 | | CAS: | 119-15-3 | | MF: | C12H9N3O5 | | MW: | 275.22 | | EINECS: | 204-300-4 | | Product Categories: | | | Mol File: | 119-15-3.mol | 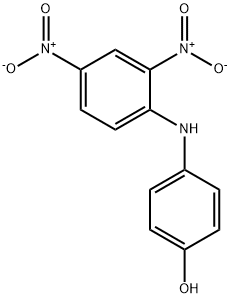 |
| | 4-(2,4-DINITROANILINO)PHENOL Chemical Properties |
| Melting point | 191 °C (dec.)(lit.) | | Boiling point | 418.17°C (rough estimate) | | density | 1.3877 (rough estimate) | | refractive index | 1.5700 (estimate) | | solubility | Chloroform (Very Slightly), DMSO (Slightly) | | form | Solid | | pka | 10.20±0.26(Predicted) | | color | Very Dark Orange | | Water Solubility | 2.477mg/L(25 ºC) | | CAS DataBase Reference | 119-15-3(CAS DataBase Reference) | | EPA Substance Registry System | 4-(2,4-Dinitroanilino)phenol (119-15-3) |
| Hazard Codes | F,Xi | | Risk Statements | 11-36/37/38 | | Safety Statements | 16-26-33-36/37/39 | | RIDADR | UN 1325 4.1/PG 2 | | WGK Germany | 2 | | RTECS | SL3070000 | | HazardClass | 4.1 | | PackingGroup | III |
| | 4-(2,4-DINITROANILINO)PHENOL Usage And Synthesis |
| Uses | Disperse Yellow 1 is an azo disperse dye which is water insoluble. Disperse Yellow 1 can be used to dye polyester and acetate fibers. Dyes and metabolites, Environmental Testing. | | Preparation | 4-Aminophenol and 1-Chloro-2,4-dinitrobenzene??condensation | | General Description | Reddish-brown solid. | | Air & Water Reactions | Insoluble in water. | | Reactivity Profile | 4-(2,4-DINITROANILINO)PHENOL is an organonitrate/phenol. Organonitrate compounds range from slight to strong oxidizing agents. If mixed with reducing agents, including hydrides, sulfides and nitrides, they may begin a vigorous reaction that culminates in a detonation. The aromatic nitro compounds may explode in the presence of a base such as sodium hydroxide or potassium hydroxide even in the presence of water or organic solvents. The explosive tendencies of aromatic nitro compounds are increased by the presence of multiple nitro groups. Phenols do not behave as organic alcohols, as one might guess from the presence of a hydroxyl (-OH) group in their structure. Instead, they react as weak organic acids. Phenols and cresols are much weaker as acids than common carboxylic acids (phenol has Ka = 1.3 x 10^[-10]). These materials are incompatible with strong reducing substances such as hydrides, nitrides, alkali metals, and sulfides. Flammable gas (H2) is often generated, and the heat of the reaction may ignite the gas. Heat is also generated by the acid-base reaction between phenols and bases. | | Fire Hazard | Flash point data for 4-(2,4-DINITROANILINO)PHENOL are not available, however, 4-(2,4-DINITROANILINO)PHENOL is probably combustible. | | Properties and Applications | yellow. Insoluble in water, slightly soluble in alcohol. In concentrated sulfuric acid for claybank, dilution after yellow. Used for cellulose ester, polystyrene and other thermoplastic resin paint color. |
| | 4-(2,4-DINITROANILINO)PHENOL Preparation Products And Raw materials |
|