| Company Name: |
TargetMol Chemicals Inc.
|
| Tel: |
4008200310 |
| Email: |
marketing@tsbiochem.com |
| Products Intro: |
Product Name:Chlornaphazine;Chlornaphazine
CAS:494-03-1
Purity:98% Package:5 mg
|
|
| | chlornaphazine Basic information |
| Product Name: | chlornaphazine | | Synonyms: | DI-(2-CHLOROETHYL)-BETA-NAPHTHYLAMINE;DICHLOROETHYL-BETA-NAPHTHYLAMINE;Naphthalenamine, NN’-bis(2-chloroethyl)-;CB 1048;Cloronaftina;Erysan;N,N-Bis(2-chloroethyl)-2-naphtylamine;R48 | | CAS: | 494-03-1 | | MF: | C14H15Cl2N | | MW: | 268.185 | | EINECS: | 2077850 | | Product Categories: | | | Mol File: | 494-03-1.mol | 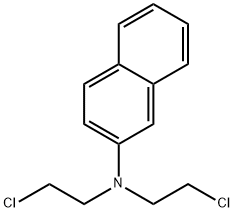 |
| | chlornaphazine Chemical Properties |
| Melting point | 54-56° | | Boiling point | bp5 210° | | density | 1.2039 (rough estimate) | | refractive index | 1.6300 (estimate) | | pka | 2.75±0.70(Predicted) | | color | Platelets from pet ether | | IARC | 1 (Vol. 4, Sup 7, 100A) 2012 | | EPA Substance Registry System | Chlornaphazine (494-03-1) |
| | chlornaphazine Usage And Synthesis |
| Description | Chlornaphazine is composed of clear, colorless plates or may be a brown solid. Molecularweight=268.2; Freezing/Melting point=54-56℃.Slightly soluble in water; solubility=, 1 mg/mL at 22℃. | | Chemical Properties | Chlornaphazine is composed of clear, colorless plates or may be a brown solid. | | Definition | ChEBI: Chlornaphazine is a member of naphthalenes. | | Brand name | Aleukon;Chloronaftina;Nafticlorina;Naphthylamine mustard. | | World Health Organization (WHO) | The World Health Organization has no information further to the
above regarding preparations containing chlornaphazine or to indicate that they
are still commercially manufactured. | | General Description | Colorless plates or brown solid. | | Air & Water Reactions | Insoluble in water. | | Reactivity Profile | chlornaphazine reacts with nucleophiles . | | Fire Hazard | Flash point data are not available for chlornaphazine , but chlornaphazine is probably combustible. | | Safety Profile | Confirmed human carcinogenproducing bladder tumors. Human and experimentalcarcinogenic data. Moderately toxic by intraperitonealroute. When heated to decomposition it emits very toxicfumes of Cl- and NOx. | | Potential Exposure | May be combustible and possibly
explosive. Not produced or used commercially in the
United States, Chlornaphazine has been used in other countries in the treatment of leukemia and related cancers.
Currently, this drug does not have wide therapeutic usage. | | First aid | Eye Contact: Remove any contact lenses at once.Immediately flush eyes well with copious quantities ofwater or normal saline for at least 20-30 min. Seek medicalattention. Inhalation: Leave contaminated area immediately; breathe fresh air. Proper respiratory protection mustbe supplied to any rescuers. If coughing, difficult breathingor any other symptoms develop, seek medical attention atonce, even if symptoms develop many hours after exposure.Ingestion: Contact a physician, hospital or poison center atonce. If the victim is unconscious or convulsing, do notinduce vomiting or give anything by mouth. Assure that thepatient’s airway is open and lay him on his side with hishead lower than his body and transport immediately to amedical facility. If conscious and not convulsing, give aglass of water to dilute the substance. Vomiting should notbe induced without a physician’s advice. | | storage | Color Code—Blue: Health Hazard/Poison: Storein a secure poison location. Prior to working with thischemical you should be trained on its proper handling andstorage. Store in a refrigerator or a cool, dry place. A regulated, marked area should be established where this chemical is handled, used, or stored in compliance with OSHAStandard 1910.1045. | | Shipping | UN2810 Toxic liquids, organic, n.o.s., Hazard
Class: 6.1; Labels: 6.1-Poisonous materials, Technical Name
Required. UN2588 Pesticides, solid, toxic, Hazard Class: 6.1;
Labels: 6.1-Poisonous materials, Technical Name Required. | | Purification Methods | Crystallise it from pet ether. At 15o it is soluble in EtOH (3.2%), Et2O (50%), Me2CO (84%) and *C6H6 (80%). [Beilstein 12 III 2996, 12 IV 3126.] CARCINOGENIC. | | Incompatibilities | Keep away from oxidizers, nucleophiles,
and nitromethanes. Amines may react violently with nitric
acid; cellulose nitrate (of high surface area); 1-chloro-2,3-epoxypropane (exothermic reaction may occur). Incompatible with
acids, acid halides; alcohols, aldehydes, alkylene oxides; anhydrides, cresols, caprolactam solution; epichlorohydrin, epoxides, isocyanates, glycols, peroxides, phenols. Contact with
strong reducing agents may generate flammable hydrogen gas.
May attack copper and copper alloys. | | Waste Disposal | Disposal of unused product
must be undertaken by qualified personnel who are knowledgeable in all applicable regulations and follow all pertinent safety precautions including the use of appropriate
protective equipment. For proper handling and disposal,
always comply with federal, state, and local regulations. |
| | chlornaphazine Preparation Products And Raw materials |
|