2-(1H-Imidazol-1-yl)ethanamine manufacturers
|
| | 2-(1H-Imidazol-1-yl)ethanamine Basic information |
| Product Name: | 2-(1H-Imidazol-1-yl)ethanamine | | Synonyms: | 2-IMIDAZOL-1-YL-ETHYLAMINE DIHYDROCHLORIDE;2-Imidazol-1-yl-ethylamine1500;1H-Imidazole-1-ethanamine 2HCl;1H-Imidazole-1-ethanamine;N-(2-Aminoethyl)imidazole;[2-(1H-imidazol-1-yl)ethyl]amine hydrochloride;2-(1H-Imidazol-1-yl)ethanamine;2-(1H-imidazol-1-yl)ethanamine(SALTDATA: 2HCl) | | CAS: | 5739-10-6 | | MF: | C5H9N3 | | MW: | 111.15 | | EINECS: | | | Product Categories: | Imidazol&Benzimidazole | | Mol File: | 5739-10-6.mol | 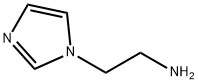 |
| | 2-(1H-Imidazol-1-yl)ethanamine Chemical Properties |
| Boiling point | 283℃ | | density | 1.15 | | Fp | 125℃ | | storage temp. | Keep in dark place,Inert atmosphere,Room temperature | | pka | 7.51±0.10(Predicted) |
| | 2-(1H-Imidazol-1-yl)ethanamine Usage And Synthesis |
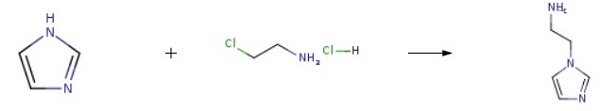
| Uses | 2-(1H-Imidazol-1-yl)ethanamine is an intermediate for the preparation of selective tumor inhibitor bis(nitroimidazolyl)alkanecarboxamide hypoxia. | | Synthesis | Imidazole (8.10 g, 119 mmol), 2-chloroethylamine monohydrochloride (15.2 g, 131 mmol), tetrabutylammonium hydrogensulfate (1.62 g, 4.8 mmol), and sodium hydroxide (17.1 g, 428 mmol) were combined with 100 ml acetonitrile and heated under reflux for 21 h. The reaction mixture was cooled and filtered. The filtrate was concentrated to a pale yellow oil. Flash chromatography (silica gel, gradient elution of acetonitrile to 9:1 acetonitrile/NH4OH) afforded 4.52 g (34%) of 2-(1H-Imidazol-1-yl)ethanamine as a pale yellow oil.
 |
| | 2-(1H-Imidazol-1-yl)ethanamine Preparation Products And Raw materials |
|