| Company Name: |
Varanous Labs Pvt Ltd
|
| Tel: |
+91-7036248882 +91-7036248882 |
| Email: |
bheemashankar.e@varanouslabs.com |
| Products Intro: |
Product Name:Erythromycin Imp F
CAS:105882-69-7
Purity:98% Package:1 kg,5 kg, 10 kg,25kg and 1 MT
|
|
| | LY 267108 Basic information |
| Product Name: | LY 267108 | | Synonyms: | LY 267108;(2R,3R,6R,7S,8S,9R,10R)-7-[(2,6-Dideoxy-3-C-Methyl-3-O-Methyl-α-L-ribo-hexopyranosyl)oxy]-3-[(1R,2R)-1,2-dihydroxy-1-Methylbutyl]-2,6,8,10,12-pentaMethyl-9-[[3,4,6-trideoxy-3-(diMethylaMino)-β-D-xylo-hexopyranosyl]oxy]-4,13-dioxabicyclo[8.2.1]tridec-12-en-5-one;8,9-Anhydropseudo-erythroMycin A-6,9-heMiketal;(2R,3R,6R,7S,8S,9R,10R)-7-[(2,6-Dideoxy-3-C-methyl-3-O-methyl-alpha-L-ribo-hexopyranosyl)oxy]-3-[(1R,2R)-1,2-dihydroxy-1-methylbutyl]-2,6,8,10,12-pentamethyl-9-[[3,4,6-trideoxy-3-(dimethylamino)-beta-D-xylo-hexopyranosyl]oxy]-4,13-dioxabicyclo[8.2.1]tridec-12-en-5-one;NMIWBQUQCOMGHJ-XWVSLMRSSA-N;Erythromycin EP Impurity F/(2R,3R,6R,7S,8S,9R,10R)-3-((2R,3R)-2,3-dihydroxypentan-2-yl)-9-(((2S,3R,4S,6R)-4-(dimethylamino)-3-hydroxy-6-methyltetrahydro-2H-pyran-2-yl)oxy)-7-(((2R,4R,5S,6S)-5-hydroxy-4-methoxy-4,6-dimethyltetrahydro-2H-pyran-2-yl)oxy)-2,6;Erythromycin Impurit F:(pseudoerythromycin A enol ether);Erythromycin Impurity 15(Erythromycin EP Impurity F) | | CAS: | 105882-69-7 | | MF: | C37H65NO12 | | MW: | 715.91 | | EINECS: | | | Product Categories: | Intermediates & Fine Chemicals;Metabolites & Impurities;Pharmaceuticals;Chromatography;Drugs&Metabolites;Forensic and Veterinary Standards;Neat CompoundsAnalytical Standards;Pharmacology Standards | | Mol File: | 105882-69-7.mol | 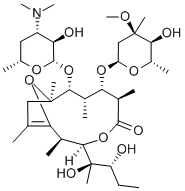 |
| | LY 267108 Chemical Properties |
| Melting point | 127.0-130.0 °C | | Boiling point | 810.7±65.0 °C(Predicted) | | density | 1.20±0.1 g/cm3(Predicted) | | storage temp. | −20°C | | solubility | DMF: Soluble; DMSO: Soluble; Ethanol: Soluble; Methanol: Soluble | | pka | 13.45±0.70(Predicted) |
| | LY 267108 Usage And Synthesis |
| Chemical Properties | White Solid | | Uses | Pseudoerythromycin A enol ether is a degradation product of erythromycin formed by a complex internal rearrangement of erythromycin A on exposure to neutral to weakly alkaline conditions. The C6–OH forms an internal enol ether with the C9 ketone of erythromycin, while the C11-OH attacks the carbonyl of the lactone to reduce the macrocycle from a 14- to an 11-membered macrolide. Synthetically, pseudoerythromycin A enol ether is prepared by reacting erythromycin enol ether with carbonate. Pseudoerythromycin A enol ether is devoid of antibiotic activity but is an important analytical standard for erythromycin A stability studies. | | Uses | Erythromycin impurity. | | Uses | Pseudo Erythromycin A Enol Ether (Erythromycin EP Impurity F) is an Erythromycin impurity. |
| | LY 267108 Preparation Products And Raw materials |
|