|
|
| | 2-Chloro-4-methyl-3-nitropyridine Basic information |
| | 2-Chloro-4-methyl-3-nitropyridine Chemical Properties |
| Melting point | 51-53 °C (lit.) | | Boiling point | 279.6±35.0 °C(Predicted) | | density | 1.406±0.06 g/cm3(Predicted) | | Fp | >230 °F | | storage temp. | Keep in dark place,Sealed in dry,Room Temperature | | solubility | Chloroform (Slightly), DMSO (Slightly), Methanol (Slightly) | | pka | -1.80±0.10(Predicted) | | form | Powder | | color | Yellow to beige | | Sensitive | Hygroscopic | | InChI | InChI=1S/C6H5ClN2O2/c1-4-2-3-8-6(7)5(4)9(10)11/h2-3H,1H3 | | InChIKey | JHARVUVBTAAPLA-UHFFFAOYSA-N | | SMILES | C1(Cl)=NC=CC(C)=C1[N+]([O-])=O | | CAS DataBase Reference | 23056-39-5(CAS DataBase Reference) |
| | 2-Chloro-4-methyl-3-nitropyridine Usage And Synthesis |
| Chemical Properties | Light yellow Cryst | | Uses | 2-Chloro-4-methyl-3-nitropyridine is a cyclopropyldipyridodiazepinone derivative for use as non-nucleoside reverse transcriptase inhibitors. | | Definition | 2-Chloro-4-methyl-3-nitropyridine (2C4M3NP) is a nitropyridine derivative that could form an acceptor fragment of 2-adamantylamino-5-nitropyridine (AANP). This crystal shows a particularly large optical non-linearity. It is observed that the mean C-C (ring) bond distance calculated between the ring carbon atoms of 2C4M3NP is 1.387 Å. In X-ray data of pyridine, the mean C-C (ring) bond distance is 1.394 Å. The C-H bond distances are found to be 1.081 Å. The computed values of C-C (ring) and C-H are slightly higher than that of the experimental value. This is attributed to the substituents present in the pyridine. The C-C-C bond angle where the -Cl and -NO2 group is attached with the middle carbon is more than 120? while the C-C-C bond angle where the -CH3 is substituted is less than 120? in 2C4M3NP. The N-C-CH3 bond angle (116.3。) is less than that of the C-C-CH3 (124.2。) probably due to the weak hydrogen bond formed between the nitrogen and hydrogen of the methyl group[1].
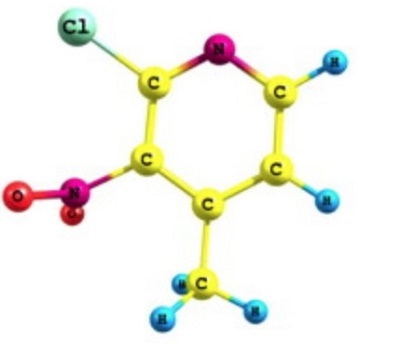 | | General Description | Experimental FTIR and FT-Raman spectroscopic analysis of 2-chloro-4-methyl-3-nitropyridine was reported. | | References | [1] Arjunan, V. , et al. "A comparative study on vibrational, conformational and electronic structure of 2-chloro-4-methyl-3-nitropyridine and 2-chloro-6-methylpyridine." Spectrochimica Acta Part A Molecular & Biomolecular Spectroscopy 92.none(2012):305-317. |
| | 2-Chloro-4-methyl-3-nitropyridine Preparation Products And Raw materials |
|