Inorganic chemicals is the shortened form of inorganic chemical industry and is an important branch of the chemical industry with natural resources and industrial by-products as raw materials for the production of sulfuric acid, nitric acid, hydrochloric acid, phosphoric acid, soda ash, caustic soda, synthetic ammonia, fertilizer and inorganic salts, etc. This includes sulfuric acid industry, soda industry, the chloro-alkali industry, synthetic ammonia industry, fertilizer industry and mineral industry. Its broad definition also includes the production of inorganic non-metallic materials and fine inorganic product such as ceramics and inorganic pigment. The main raw material of inorganic chemical products are mineral product including sulfur, sodium, phosphorus, potassium and calcium and coal, oil, gas, and air, water and so on. Inorganic chemicals can be traced back to the ancient process of ceramics, alchemy, brewing, dyeing at thousands of years ago. Although with small scale, backward technology and pure manual manipulation, but it is the prototype of inorganic chemicals. For thousands of years, due to the low productivity, it gets slow development. Until the 18th century, it had developed rapidly. In the middle of 18th century, Britain had first applied lead chamber method using saltpeter and sulfur as raw materials to produce sulfuric acid. In 1783, Lu Bulan (France) proposed the soda method using sodium chloride, sulfuric acid, coal as raw materials. In the latter half of the 18th century, the modern chemical industry taking inorganic chemical industry as the main content had began to emerge. In 1841, people began the production of phosphate fertilizer; In 1965 Belgian Solvay realized the industrialization of ammonia soda for production of soda; with the rise of preparing potassium industry in 1870; In 1890, people began to use electrolytic approach for making Cl2 and caustic soda; In 1913, people had achieved the catalytic synthesis
How are Diamonds Formed?
For centuries, diamonds have been regarded as a gemstone with extraordinary brilliance and luster. This article will introduce how the diamonds formed in nature and how to make it in laboratory.
Mar 8,2024 Inorganic chemistryIs silver chloride soluble in water? is it used for?
Silver chloride is a white crystalline solid that is well known for its low solubility in water and its sensitivity to light.
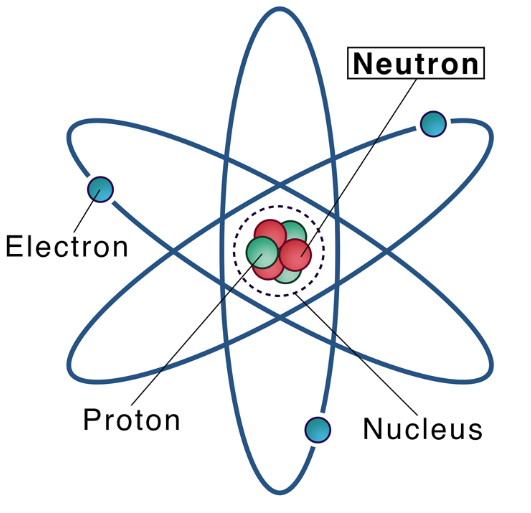
Mar 7,2024 Inorganic chemistryHow many neutrons, protons and electrons does a sodium atom have?
A neutron is a neutral subatomic particle that, in conjunction with protons, makes up the nucleus of every atom except ordinary hydrogen (whose nucleus has one proton and no neutrons).
Mar 6,2024 Inorganic chemistryIs silicon dioxide in supplements and food safe?
Silicon dioxide, also known as silica or SiO2, is a naturally occurring compound. It's made of silicon and oxygen.
Mar 6,2024 Inorganic chemistryHow long does it take for boric acid to dissolve?
Understanding the dissolution process is crucial to ensure the suppositories are effective and provide relief. Boric acids take between 4-12 hours to dissolve. Avoid any intercourse till the boric aci
Mar 6,2024 Inorganic chemistrySodium hydroxide:pH calculation,Uses,Health hazards
Liquid sodium hydroxide is colorless and has no odor. It can react violently with strong acids and with water.
Mar 6,2024 Inorganic chemistryThe uses of Boric acid
Boric acid is a water-soluble white compound and occurs naturally. Its molecular formula is H3BO3.
Mar 5,2024 Inorganic chemistryIs uranium dioxideperoxide safe?
Uranium dioxideperoxide is very dangerous! The substance is fatal if swallowed, is fatal if inhaled, is toxic to aquatic life with long lasting effects and may cause damage to organs through prolonged
Mar 4,2024 Inorganic chemistryThe Application of Nickel Carbonate as Catalyst
Nickel carbonate finds applications in sulphamate baths, metal phosphating, electroplating, and ceramic processes. This article will focus on its utilization as a catalyst.
Mar 4,2024 Inorganic chemistryWhat is the compound name for (NH4)3PO4?
The name of the compound (NH4)3PO4 is Ammonium Phosphate. Ammonium phosphate is an unstable compound made of ammonium and phosphate salt with the chemical formula (NH4)3PO4.
Mar 4,2024 Inorganic chemistry