| Identification | Back Directory | [Name]
1,4-Bis(diphenylphosphino)butane-palladium(II) chloride | [CAS]
29964-62-3 | [Synonyms]
PdCl2(dppb)
Palladium(ii) chloride 1,4-b
[(C6H5)2PCH2CH2CCH2H2P(C6H5)2]PdCl2
Bisdiphenylphosphinobutanepalladiumdichloride
4-Bis(diphenylphosphino)butane-palladiuM(II) chloride
Palladium(II)chloride-1,4-bis(diphenylphosphine)butane
1,4-BIS(DIPHENYLPHOSPHINE)BUTANE PALLADIUM(II) CHLORIDE
1,4-BIS(DIPHENYLPHOSPHINO)BUTANE-PALLADIUM(II) CHLORIDE
DICHLORO[1,4-BIS(DIPHENYLPHOSPHINO)BUTANE]PALLADIUM(II)
1,4-Butylenebis(diphenylphosphine)-palladium dichloride
1,4-bis (diphenylphosphino butane) palladiuM dichloride
[1,4-BIS(DIPHENYLPHOSPHINO)BUTANE]PALLADIUM(II) DICHLORIDE
1,4- double(twophenyl phosphinebutane)twopalladiuM chloride
1,4-Bis(diphenylphosphino)butane-palladium(II) chloride,98+%
Dichloro[1,4-bis(diphenylphosphino)butane]palladium (II) ,98%
dichloropalladium,4-diphenylphosphanylbutyl(diphenyl)phosphane
PALLADIUM(II) CHLORIDE 1,4-BIS(DIPHENYLPHOSPHINO)BUTANE COMPLEX
Dichloro[bis(1,4-diphenylphosphino)butane]palladium(II), Pd 17.6%
Dichloro-1,4-bis(diphenylphosphino)butane-palladium(II),PdCl2(DPPB)
PDCL2(DPPB)/ 1,4-BIS(DIPHENYLPHOSPHINE)BUTANE PALLADIUM(II)CHLORIDE
Dichloro 1,4-bis(diphenylphosphino)butane palladium (II) For use in coupling reactions
1,4-Bis(diphenylphosphino)butane-palladium(II) chloride
Dichloro-1,4-bis(diphenylphosphino)butane-palladium(II)
Dichloro-1,4-bis(diphenylphosphino)butane-palladium(II), Palladium(II) chloride 1,4-bis(diphenylphosphino)butane complex | [EINECS(EC#)]
243-231-4 | [Molecular Formula]
C28H28P2.Cl2Pd | [MDL Number]
MFCD02093437 | [MOL File]
29964-62-3.mol | [Molecular Weight]
603.8 |
| Chemical Properties | Back Directory | [Appearance]
yellow solid | [Melting point ]
285-305 °C | [storage temp. ]
Inert atmosphere,2-8°C | [solubility ]
Soluble in ethanol, benzene, and ethereal solvents. | [form ]
Powder | [color ]
light-yellow | [InChI]
1S/C28H28P2.2ClH.Pd/c1-5-15-25(16-6-1)29(26-17-7-2-8-18-26)23-13-14-24-30(27-19-9-3-10-20-27)28-21-11-4-12-22-28;;;/h1-12,15-22H,13-14,23-24H2;2*1H;/q;;;+2/p-2 | [InChIKey]
JQXJBXVWVPVTOO-UHFFFAOYSA-L | [SMILES]
P(C1=CC=CC=C1)(CCCCP(C1=CC=CC=C1)C1C=CC=CC=1)C1=CC=CC=C1.[Pd](Cl)Cl |
| Hazard Information | Back Directory | [Chemical Properties]
yellow solid | [Uses]
Dichloro[bis(1,4-diphenylphosphino)butane]palladium(II) is used for the catalysis of styrene carbonylation, coupling of alkyl Grignard reagents with organic halides, selective monoalkylation of organic polyhalides, and modification of the dihalovinyl moiety of synthetic pyrethroids. | [Preparation]
Dichloro[1,4-bis(diphenylphosphino)butane]palladium(II) (1,4-Bis(diphenylphosphino)butane-palladium(II) chloride) can be prepared conveniently in situ by mixing the bidentate phosphine ligand and
(PhCN)2PdCl2 in either benzene or chloroform.[1] The preparation of (dppb)PdCl2 is also possible through the
reaction of PdCl2 with dppb in acetone, a process that slowly forms the catalyst as a white precipitate which
can be isolated in 51% yield.[2] Preparation of the hydrate complex is possible via the reaction of Na2[PdCl4]
with dppb in a mixture of CH2Cl2 and water. | [reaction suitability]
core: palladium
reaction type: Buchwald-Hartwig Cross Coupling Reaction
reaction type: Cross Couplings
reaction type: Heck Reaction
reaction type: Hiyama Coupling
reaction type: Negishi Coupling
reaction type: Sonogashira Coupling
reaction type: Stille Coupling
reaction type: Suzuki-Miyaura Coupling
reagent type: catalyst | [Synthesis]
Example 3
Step 1: 10 mL of palladium sponge was mixed with concentrated hydrochloric acid and concentrated nitric acid in a 3:1 volume ratio until the palladium sponge was completely dissolved. 37% concentrated hydrochloric acid was added to the solution and filtered to remove insoluble material while treating the brown nitrogen oxides produced. The filtrate was concentrated to a solution of chloropalladium acid with a palladium concentration of 0.5 g/mL.
Step 2: An N,N-dimethylformamide solution of bis(diphenylphosphino)butane was prepared by adding 44 g of 1,4-bis(diphenylphosphino)butane to 100 mL of N,N-dimethylformamide, and heating to 50°C. The N,N-dimethylformamide solution of bis(diphenylphosphino)butane was then heated to 50°C.
Step 3: The chloropalladium acid solution prepared in step 1 was added dropwise to the bis(diphenylphosphino)butane solution in step 2 under stirring. The reaction was stirred at 50 °C for 60 minutes. After completion of the reaction, the reaction solution was cooled and filtered, and the filter cake was collected.
Step 4: The filter cake obtained in Step III was washed with anhydrous ethanol and then dried under vacuum at 50 °C for 4 h. A light yellow 1,4-bis(diphenylphosphino)butane-palladium(II) chloride complex (C28H28Cl2P2Pd) of 56.1 g was obtained in 98.9% yield. | [References]
1. Sugi, Y.; Bando, K.-I. CL 1976, 727.
2. Sanger, A. R. JCS(D) 1977, 1971.
|
| Questions And Answer | Back Directory | [Reaction]
- Catalyst for the Grignard coupling for regioand stereoselective monoalkylation and arylation of 1,1-dichloro-1-alkenes
- Synthetic pyrethriods: catalyst for the Negishi coupling of (2,2-dihaloethenyl)cyclopropanecarboxylates
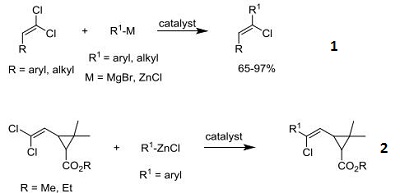
|
|
|