34107-46-5
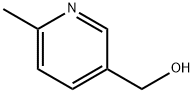 34107-46-5 结构式
34107-46-5 结构式
基本信息
6-甲基-3-甲醇吡啶
6-甲基-3-羟甲基吡啶
(6-甲基-3-吡啶基)甲醇
(6-甲基吡啶-3-基)甲醇
2-甲基-5-(羟甲基)吡啶
6-Methylpyridine-3-methanol
6-Methyl-3-PyridineMethanol
(6-Methyl-3-pyridyl)methanol
6-(Methylpyridin-3-yl)methano
3-Pyridinemethanol, 6-methyl-
(6-methyl-3-pyridinyl)methanol
6-Methyl-3-pyridinemethanol >
5-(Hydroxymethyl)-2-methylpyridine
5-Hydroxymethyl-2-methylpyridine 97%
物理化学性质
安全数据
制备方法
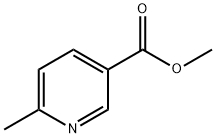
5470-70-2

34107-46-5
以6-甲基烟酸甲酯为原料合成6-甲基-3-甲醇吡啶的一般步骤:在-5℃下,向搅拌的6-甲基烟酸甲酯(21g; 140mmol; 1当量)的无水THF(150mL)溶液中缓慢滴加1M LiAlH4的THF溶液(210mL; 210mmol; 1.5当量)。滴加完毕后,将反应混合物在23℃下继续搅拌2小时。随后,将混合物冷却至-10℃,并用硫酸钠十水合物和乙酸乙酯小心淬灭反应,直至停止产生气泡。将混合物通过硅藻土垫过滤,滤液经减压浓缩至干,得到6-甲基-3-甲醇吡啶(17g,产率100%)。产物经1H NMR(DMSO-d6)表征:δ 8.37(s,1H),7.59(dd,1H,J = 2,8Hz),7.19(d,1H,J = 8Hz),5.22(t,1H,J = 6Hz),4.47(d,2H,J = 6Hz),2.45(s,3H)。
参考文献:
[1] Patent: WO2015/95128, 2015, A1. Location in patent: Paragraph 0152; 0155; 0156
[2] Organic Preparations and Procedures International, 2015, vol. 47, # 3, p. 220 - 226
[3] Patent: US2005/124586, 2005, A1. Location in patent: Page/Page column 7
[4] Journal of the Chemical Society. Perkin Transactions 2, 2002, # 5, p. 928 - 937
[5] Patent: WO2018/165718, 2018, A1. Location in patent: Page/Page column 100; 101; 102

