3652-89-9
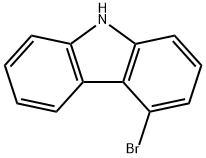 3652-89-9 结构式
3652-89-9 结构式
基本信息
4-溴-9H-咔唑
4-溴咔唑 10G
4-broMocarbazole
Carbazole, 4-bromo-
4-bromo-1H-carbazole
4-Bromocarbazole >
4-BroMo-9H-carbazole
9H-Carbazole, 4-broMo-
4-BroMo-9H-carbazole (4BC)
1-CYCLOPROPYL-2,6-PIPERAZINEDIONE
4-BroMo-9H-carbazole ISO 9001:2015 REACH
物理化学性质
制备方法
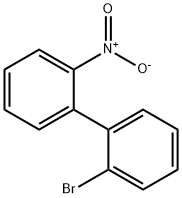
17613-47-7

3652-89-9
以2-溴-2'-硝基-1,1'-联苯为原料合成4-溴-9H-咔唑的一般步骤:将2-溴-2'-硝基-1,1'-联苯(4.7 g,16.9 mmol)和三苯基膦(11.08 g,42.25 mmol)溶解于1,2-二氯苯(110 mL)中。将反应混合物加热回流12小时,随后冷却至室温。反应液用二氯甲烷和水进行萃取,合并有机层并用无水硫酸镁干燥。减压浓缩后,通过柱色谱法(洗脱剂:二氯甲烷/正己烷,1:9,v/v)纯化,得到白色粉末状产物4-溴-9H-咔唑3.6 g,产率87%。产物表征数据如下:1H NMR (200 MHz, CDCl3) δ 8.75 (d, 1H, J = 8.0 Hz), 8.18 (s, 1H), 7.49-7.22 (m, 6H); MS (FAB) m/z 247 [(M + H)+]。
参考文献:
[1] Organic Electronics: physics, materials, applications, 2013, vol. 14, # 1, p. 67 - 73
[2] Patent: WO2017/196081, 2017, A1. Location in patent: Paragraph 281; 282; 283; 284
[3] Patent: KR2016/6007, 2016, A. Location in patent: Paragraph 0439-0441
[4] Patent: KR2016/20159, 2016, A. Location in patent: Paragraph 0356-0359
[5] Patent: KR2015/133097, 2015, A. Location in patent: Paragraph 0282; 0283
