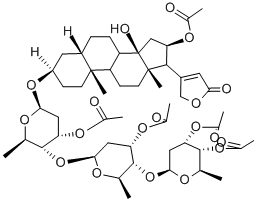
Pengitoxin
- CAS No.
- 7242-04-8
- Chemical Name:
- Pengitoxin
- Synonyms
- Pentaacetate;pentaacetylgitoxin;Card-20(22)-enolide, 16-(acetyloxy)-3-[(O-3,4-di-O-acetyl-2,6-dideoxy-β-D-ribo-hexopyranosyl-(1→4)-O-3-O-acetyl-2,6-dideoxy-β-D-ribo-hexopyranosyl-(1→4)-3-O-acetyl-2,6-dideoxy-β-D-ribo-hexopyranosyl)oxy]-14-hydroxy-, (3β,5β,16β)-
- CBNumber:
- CB11118358
- Molecular Formula:
- C51H74O19
- Molecular Weight:
- 991.12
- MDL Number:
- MOL File:
- 7242-04-8.mol
| Melting point | 151-155° |
|---|---|
| alpha | D20 +14.0 ±1.5° (c = 1.6 in pyridine) |
| Boiling point | 742.94°C (rough estimate) |
| Density | 1.1187 (rough estimate) |
| refractive index | 1.6500 (estimate) |
| Water Solubility | 11.89mg/L(temperature not stated) |
| FDA UNII | 15RPL803R9 |
SAFETY
Risk and Safety Statements
| Toxicity | LD50 in mice (mg/kg): 6.4 i.p.; in rats (mg/kg): 21.0 i.v. (Foerster) |
|---|
Pengitoxin Chemical Properties,Uses,Production
Originator
Pengitoxin,Shanghai Lansheng
Manufacturing Process
Gitoxin is isolated from leaves of Digitalis purpurea L. genus Scrofphulariaceae.
10.0 g pure gitoxin are boiled under reflux with 1 L pure acetic anhydride. Gitoxin thereby goes into solution in the course of 1 h. The boiling is discontinued after 1 h and the acetic anhydride distilled off in a vacuum as completely as possible. After taking up the oily residue with 500 ml chloroform, the solution is successively washed with 200 ml of 2 N potassium bicarbonate solution, 0.1 N hydrochloric acid and water. After drying the chloroform solution with anhydrous sodium sulfate, the chloroform is first distilled off over an open flame and then on a water bath at 40°C in a vacuum, a substantially crystalline residue thereby being obtained. The 12.8 g of crude product obtained are recrystallized from 12 times the amount of a mixture of pyridine, methanol and water (25:10:65) to give rhombic crystals, melting point 151°-155°C.
Therapeutic Function
Cardiotonic
Pengitoxin Preparation Products And Raw materials
Pengitoxin Suppliers
| Supplier | Tel | Country | ProdList | Advantage | |
|---|---|---|---|---|---|
| 2A PharmaChem USA | -- | sales@2apharmachem.com | United States | 6148 | 39 |
| 3B Scientific Corporation | -- | sales@3bsc.com | United States | 6744 | 47 |
| Supplier | Advantage |
|---|---|
| 2A PharmaChem USA | 39 |
| 3B Scientific Corporation | 47 |