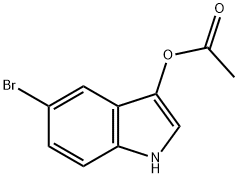
3-ACETOXY-5-BROMOINDOLE
- CAS No.
- 17357-14-1
- Chemical Name:
- 3-ACETOXY-5-BROMOINDOLE
- Synonyms
- 5-BROMOINDOXYL ACETATE;5-BROMOINDOLYL ACETATE;O-ACETYL-5-BROMOINDOXYL;3-ACETOXY-5-BROMOINDOLE;5-BROMOINDOXYL-3-ACETATE;5-BROMOINDOLYL 3-ACETATE;5-Bromo-3-indoxylacetate;5-Bromo-3-indolyl Acetate;5-Bromoindoxyl acetate,98%;5-Bromoindoxyl Acetate >
- CBNumber:
- CB2103635
- Molecular Formula:
- C10H8BrNO2
- Molecular Weight:
- 254.08
- MDL Number:
- MFCD00037933
- MOL File:
- 17357-14-1.mol
- MSDS File:
- SDS
- TDS File:
- TDS
| Product description | Number | Pack Size | Price |
| 5-Bromoindoxyl acetate 98% | 374210 | 1g | $388 |
| 5-Bromoindoxyl Acetate >98.0%(HPLC)(N) | A0940 | 100mg | $41 |
| 3-Acetoxy-5-bromoindole | FA52314 | 250mg | $55 |
| 3-Acetoxy-5-bromoindole | 5823AF | 100mg | $84 |
| 3-Acetoxy-5-bromoindole | FA52314 | 500mg | $90 |
| More product size | |||
| Melting point | 130-132 °C(lit.) |
|---|---|
| storage temp. | Keep in dark place,Inert atmosphere,Room temperature |
| solubility | ethanol: soluble50mg/mL, clear, colorless |
| form | powder to crystal |
| color | White to Purple |
| BRN | 168698 |
| InChI | 1S/C10H8BrNO2/c1-6(13)14-10-5-12-9-3-2-7(11)4-8(9)10/h2-5,12H,1H3 |
| InChIKey | KFTGECHXNQBTNZ-UHFFFAOYSA-N |
| SMILES | CC(=O)Oc1c[nH]c2ccc(Br)cc12 |
| CAS DataBase Reference | 17357-14-1(CAS DataBase Reference) |
| FDA UNII | FBY7EQC6N4 |
| EPA Substance Registry System | 1H-Indol-3-ol, 5-bromo-, acetate (ester) (17357-14-1) |
| UNSPSC Code | 12352100 |
| NACRES | NA.22 |
SAFETY
Risk and Safety Statements
| Symbol(GHS) |  GHS07 |
|---|---|
| Signal word | Warning |
| Hazard statements | H302-H317 |
| Precautionary statements | P280-P305+P351+P338 |
| PPE | Eyeshields, Gloves, type N95 (US) |
| Safety Statements | 22-24/25 |
| WGK Germany | 3 |
| F | 8-10 |
| TSCA | TSCA listed |
| HS Code | 2933998090 |
| Storage Class | 11 - Combustible Solids |
3-ACETOXY-5-BROMOINDOLE price More Price(28)
| Manufacturer | Product number | Product description | CAS number | Packaging | Price | Updated | Buy |
|---|---|---|---|---|---|---|---|
| Sigma-Aldrich | 374210 | 5-Bromoindoxyl acetate 98% | 17357-14-1 | 1g | $388 | 2026-04-30 | Buy |
| TCI Chemical | A0940 | 5-Bromoindoxyl Acetate >98.0%(HPLC)(N) | 17357-14-1 | 100mg | $41 | 2026-04-30 | Buy |
| Biosynth Carbosynth | FA52314 | 3-Acetoxy-5-bromoindole | 17357-14-1 | 250mg | $55 | 2021-12-16 | Buy |
| AK Scientific | 5823AF | 3-Acetoxy-5-bromoindole | 17357-14-1 | 100mg | $84 | 2021-12-16 | Buy |
| Biosynth Carbosynth | FA52314 | 3-Acetoxy-5-bromoindole | 17357-14-1 | 500mg | $90 | 2021-12-16 | Buy |
3-ACETOXY-5-BROMOINDOLE Chemical Properties,Uses,Production
Uses
5-Bromoindoxyl acetate may be used:
- as esterase substrate to investigate the activity of non-specific esterase in the matrix of developing bovine enamel
- in histochemical studies of the nonspecific esterase of mouse epididymis
- as substrate to investigate the esterase activity in guinea-pig thyroid and mouse epididymis epithelial cells
- in staining procedure for the frozen sections of muscle fixed in buffered formaldehyde
General Description
5-Bromoindoxyl acetate is a halogenated heterocyclic compound. It is widely used as esterase substrate.
Synthesis
Method I.
The 3-hydroxyindole acetate, N-bromosuccinimide and 150 mL of carbon tetrachloride. It was added into 250 mL round bottom flask and refluxed in oil bath for 2 h. The reaction process was monitored by TLC. At the completion of the reaction, the solution changed from dark yellow to light yellow and a white solid (succinimide) was precipitated. The filtrate was withdrawn and washed, and the filtrate was collected and evaporated to obtain 5-bromo-3-hydroxyindole acetate.
Method II.
In a 500 mL single-necked flask, 3-hydroxyindole acetic acid ester was dissolved in tetrahydrofuran solution, N-bromosuccinimide was added in an ice bath, and the reaction was stirred at 0C for 3 h. The reaction was completed. The reaction mixture was poured into 150 mL of ice water and extracted with methyl tert-butyl ether (100 mL4), the organic phases were combined, dried with anhydrous sodium sulfate, and concentrated to obtain 5-bromo-3-hydroxyindole acetate.
3-ACETOXY-5-BROMOINDOLE Preparation Products And Raw materials
Raw materials
Preparation Products
| Supplier | Tel | Country | ProdList | Advantage | |
|---|---|---|---|---|---|
| ATK CHEMICAL COMPANY LIMITED | +undefined-21-51877795 | ivan@atkchemical.com | China | 33024 | 60 |
| career henan chemical co | +86-0371-86658258 +8613203830695 | factory@coreychem.com | China | 29794 | 58 |
| Guangzhou Yuheng Pharmaceutical Technology Co., Ltd | +8613580539051 | joe@yuhengpharm.com | CHINA | 21142 | 58 |
| Zhengzhou Alfa Chemical Co.,Ltd | +8618530059196 | sale04@alfachem.cn | China | 9545 | 58 |
| Hefei TNJ Chemical Industry Co.,Ltd. | +86-0551-65418671 +8618949823763 | sales@tnjchem.com | China | 34563 | 58 |
| Shanghai UCHEM Inc. | +862156762820 +86-13564624040 | sales@myuchem.com | China | 8736 | 58 |
| Dayang Chem (Hangzhou) Co.,Ltd. | +86-0571-88938639 +8617705817739 | info@dycnchem.com | China | 52705 | 58 |
| PT CHEM GROUP LIMITED | +86-85511178; | peter68@ptchemgroup.com | China | 35425 | 58 |
| GIHI CHEMICALS CO.,LIMITED | +86-571-86217390; +8618058761490 | info@gihichemicals.com | China | 49968 | 58 |
| HANGZHOU LEAP CHEM CO., LTD. | +86-571-87711850 | market18@leapchem.com | China | 43333 | 58 |
View Lastest Price from 3-ACETOXY-5-BROMOINDOLE manufacturers
| Image | Update time | Product | Price | Min. Order | Purity | Supply Ability | Manufacturer | |
|---|---|---|---|---|---|---|---|---|
 |
2020-02-01 | 3-ACETOXY-5-BROMOINDOLE
17357-14-1
|
US $3.00 / KG | 1KG | 98% | 100KG | Career Henan Chemical Co |
-

- 3-ACETOXY-5-BROMOINDOLE
17357-14-1
- US $3.00 / KG
- 98%
- Career Henan Chemical Co
17357-14-1(3-ACETOXY-5-BROMOINDOLE)Related Search:
1of4