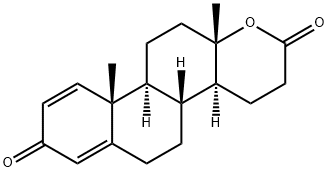
Testolactone
- CAS No.
- 968-93-4
- Chemical Name:
- Testolactone
- Synonyms
- teslak;teslac;teolit;sq9538;nsc-12173;NSC-23759;fludestrin;testolacton;TESTOLACTONE;Δ1-Testolactone
- CBNumber:
- CB4371689
- Molecular Formula:
- C19H24O3
- Molecular Weight:
- 300.39
- MDL Number:
- MFCD00866295
- MOL File:
- 968-93-4.mol
| Melting point | 218-219° |
|---|---|
| alpha | D23 -45.6° (c = 1.24 in chloroform) |
| Boiling point | 482.0±45.0 °C(Predicted) |
| Density | 1.17±0.1 g/cm3(Predicted) |
| solubility | Chloroform (Sparingly), Methanol (Slightly) |
| form | Solid |
| color | White to Off-White |
| CAS DataBase Reference | 968-93-4(CAS DataBase Reference) |
| FDA UNII | 6J9BLA949Q |
| NCI Drug Dictionary | Teslac |
Testolactone price
Testolactone Chemical Properties,Uses,Production
Originator
Fludestrin,Heyden,W. Germany,1968
Uses
Antineoplastic; non-selective steroidal aromatase inhibitor that prevents the conversion from androgen to estrogen. Used in in the treatment of familial male precocious puberty. An effective anti-tumor agent.
Uses
Testolactone USP (Teslac) is used to treat Breast cancer.
Definition
ChEBI: Testolactone is a seco-androstane and a 3-oxo-Delta(1),Delta(4)-steroid.
Manufacturing Process
(a) Fermentation: A medium of the following composition is prepared: 3.0
grams cornsteep liquor solids; 3.0 grams NH4H2PO4; 2.5 grams CaCO3; 2.2
grams soybean oil; 0.5 gram progesterone and distilled water to make 1 liter.
The medium is adjusted to pH 7.00.1. Then, 100 ml portions of the medium
are distributed in 500 ml Erlenmeyer flasks and the flasks plugged with cotton
and sterilized in the usual manner (i.e., by autoclaving for 30 minutes at
120°C). When cool, each of the flasks is inoculated with 5 to 10% of a
vegetative inoculum of Cylindrocarpon radicola [the vegetative inoculum being
grown from stock cultures (lyophilized vial or agar slant) for 48 to 72 hours in
a medium of the following composition: 15 grams cornsteep liquor; 10 grams
brown sugar; 6 grams NaNO3; 0.001 gram ZnSO4; 1.5 grams KH2PO4;0.5
gram MgSO47H2O; 5 grams CaCO3; 2 grams lard oil; and distilled water to
make 1 liter].
The flasks are then placed on a reciprocating shaker (120 one and one-half
inch cycles per minute) and mechanically shaken at 25°C for 3 days. The
contents of the flasks are then pooled and, after the pH of the culture is
adjusted to about 40.2 with sulfuric acid, filtered through Seitz filter pads to
separate the mycelium from the fermented medium.
(b) Extraction: 40 liters of the culture filtrate obtained in (a) is extracted with
40 liters chloroform in an extractor (e.g., Podbelniak, US Patent 2,530,886, or
improvements thereon) and the filtered chloroform extract is evaporated to
dryness in vacuo. The residue (11.1 grams) is taken up in 200 ml of 80%
aqueous methanol, and the resulting solution is extracted four times with 100
ml portions of hexane. The 80% aqueous methanol solution is then
concentrated in vacuo until crystals appear; and, after cooling at 0°C for
several (usually about 3 to 4) hours, the crystals formed are recovered by
filtration. About 2.9 grams 1-dehydrotestololactone (MP 217° to 217.5°C) are
thus obtained. Concentration of the mother liquors yields additionally about
6.0 grams of the lactone. Recrystallization from acetone yields a purified 1-dehydrotestololactone having a melting point of 218° to 219°C.
Therapeutic Function
Cancer chemotherapy
General Description
Testolactone, 13-hydroxy-3-oxo-13,17-secoandrosta-1,4-dien-17-oic acidΔ-lactone (Teslac),was originally synthesized as a possible anabolic steroid,considering its structural similarity to testosterone. The keystructural difference from anabolic steroids is the D-ring lactone instead of the typical cyclopentyl ring. Althoughconsidered in many texts an androgen or anabolic steroid (itis a Schedule III drug because of its classification as an anabolicsteroid), testolactone lacks androgenic effects in vivo.Its action is believed to be caused by irreversible inhibitionof aromatase. It is a relatively weak inhibitor of aromatase,but the irreversible nature of the inhibition can lead to prolongedeffects. Its relatively weak inhibition of aromataseand its undesirable dosage schedule (5×50-mg tabletsq.i.d.) give this older agent only limited use in breast cancertreatment because of better available options.
Testolactone Preparation Products And Raw materials
| Supplier | Tel | Country | ProdList | Advantage | |
|---|---|---|---|---|---|
| Hebei Mojin Biotechnology Co., Ltd | +86 13288715578 +8613288715578 | sales@hbmojin.com | China | 12446 | 58 |
| Nantong Guangyuan Chemicl Co,Ltd | +undefined17712220823 | admin@guyunchem.com | China | 616 | 58 |
| Hebei shuoxi biotechnology co. LTD | +8613081092107 | CHINA | 968 | 58 | |
| CONIER CHEM AND PHARMA LIMITED | +8618523575427 | sales@conier.com | China | 49392 | 58 |
| career henan chemical co | +86-0371-86658258 +8613203830695 | factory@coreychem.com | China | 29822 | 58 |
| Hefei TNJ Chemical Industry Co.,Ltd. | +86-0551-65418671 +8618949823763 | sales@tnjchem.com | China | 34571 | 58 |
| Wuhan Aoliqisi New Material Technology Co., Ltd. | +86-13545906766; +8613545906766 | sales@whaop.com | China | 265 | 58 |
| Hebei Qige Biological Technology Co. Ltd | +86 +8618733132031 | CHINA | 1313 | 58 | |
| Wuhan Golt Biotech Co., Ltd. | +8615389281203 | maria@goltbiotech.com | China | 980 | 58 |
| LEAP CHEM CO., LTD. | +86-852-30606658 | market18@leapchem.com | China | 24738 | 58 |
View Lastest Price from Testolactone manufacturers
| Image | Update time | Product | Price | Min. Order | Purity | Supply Ability | Manufacturer | |
|---|---|---|---|---|---|---|---|---|
 |
2024-03-08 | Testolactone
968-93-4
|
US $10.00 / kg | 1kg | 99% | 1000kg | Nantong Guangyuan Chemicl Co,Ltd | |
 |
2023-11-21 | Testolactone
968-93-4
|
US $1.00 / g | 1000g | 99% | 20ton/month | Wuhan Aoliqisi New Material Technology Co., Ltd. | |
 |
2023-09-25 | Testolactone
968-93-4
|
US $100.00-80.00 / kg | 1kg | 99% | 3 ton | Anhui Yisheng Technology Co., LTD |
-

- Testolactone
968-93-4
- US $10.00 / kg
- 99%
- Nantong Guangyuan Chemicl Co,Ltd
-

- Testolactone
968-93-4
- US $1.00 / g
- 99%
- Wuhan Aoliqisi New Material Technology Co., Ltd.
-

- Testolactone
968-93-4
- US $100.00-80.00 / kg
- 99%
- Anhui Yisheng Technology Co., LTD