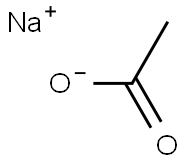
Sodium acetate
- CAS No.
- 127-09-3
- Chemical Name:
- Sodium acetate
- Synonyms
- Sodium acetate anhydrous;Anhydrous sodium acetate;BUFFER SOLUTION;Natriumacetat;sodium ethanoate;SODIUM ACETATE BUFFER;NA ACETATE;sodiiacetas;femanumber3024;natriumaceticum
- CBNumber:
- CB1230044
- Molecular Formula:
- C2H3NaO2
- Molecular Weight:
- 82.03379
- MOL File:
- 127-09-3.mol
- MSDS File:
- SDS
- Modify Date:
- 2024/5/28 19:59:05
| Melting point | >300 °C (dec.)(lit.) |
|---|---|
| Density | 1.01 g/mL at 20 °C |
| FEMA | 3024 | SODIUM ACETATE |
| refractive index | 1.4640 |
| Flash point | >250 °C |
| storage temp. | Store at +5°C to +30°C. |
| solubility | H2O: 3 M at 20 °C, clear, colorless |
| pka | 4.756[at 20 ℃] |
| form | powder |
| color | white |
| Specific Gravity | 1.45 |
| Odor | Slight acetic acid |
| PH Range | 8.5 - 9.9 at 246 g/l at 25 °C |
| PH | 7.87(1 mM solution);8.33(10 mM solution);8.75(100 mM solution);9.04(1000 mM solution) |
| Odor Type | odorless |
| Water Solubility | 500 g/L (20 ºC) |
| Sensitive | Hygroscopic |
| Hydrolytic Sensitivity | 0: forms stable aqueous solutions |
| λmax |
λ: 260 nm Amax: 0.03 λ: 280 nm Amax: 0.02 |
| Merck | 14,8571 |
| BRN | 3595639 |
| Boiling point | >400°C(decomposition) |
| Stability | Stable. Incompatible with strong oxidizing agents, halogens. Moisture sensitive. |
| InChIKey | VMHLLURERBWHNL-UHFFFAOYSA-M |
| LogP | -3.72 |
| CAS DataBase Reference | 127-09-3(CAS DataBase Reference) |
| NIST Chemistry Reference | Sodium ethanoate(127-09-3) |
| EPA Substance Registry System | Sodium acetate (127-09-3) |
SAFETY
Risk and Safety Statements
| Symbol(GHS) |  GHS07 |
|||||||||
|---|---|---|---|---|---|---|---|---|---|---|
| Signal word | Warning | |||||||||
| Hazard statements | H320 | |||||||||
| Precautionary statements | P264-P337+P313-P305+P351+P338 | |||||||||
| Safety Statements | 22-24/25 | |||||||||
| WGK Germany | 1 | |||||||||
| RTECS | AJ4300010 | |||||||||
| F | 3 | |||||||||
| Autoignition Temperature | 607 °C | |||||||||
| TSCA | Yes | |||||||||
| HS Code | 29152200 | |||||||||
| Toxicity | LD50 orally in Rabbit: 3530 mg/kg LD50 dermal Rabbit > 10000 mg/kg | |||||||||
| NFPA 704 |
|
Sodium acetate price More Price(78)
| Manufacturer | Product number | Product description | CAS number | Packaging | Price | Updated | Buy |
|---|---|---|---|---|---|---|---|
| Sigma-Aldrich(India) | W302406 | Sodium Acetate Anhydrous >99%, FCC, FG | 127-09-3 | 1SAMPLE-K | ₹5141.88 | 2022-06-14 | Buy |
| Sigma-Aldrich(India) | W302406 | Sodium Acetate Anhydrous >99%, FCC, FG | 127-09-3 | 1KG | ₹10175.5 | 2022-06-14 | Buy |
| Sigma-Aldrich(India) | W302406 | Sodium Acetate Anhydrous >99%, FCC, FG | 127-09-3 | 10KG | ₹22299.5 | 2022-06-14 | Buy |
| Sigma-Aldrich(India) | W302406 | Sodium Acetate Anhydrous >99%, FCC, FG | 127-09-3 | 25KG | ₹51862.58 | 2022-06-14 | Buy |
| Sigma-Aldrich(India) | S8750 | Sodium acetate anhydrous, ReagentPlus?, ≥99.0% | 127-09-3 | 250G | ₹3366.58 | 2022-06-14 | Buy |
Sodium acetate Chemical Properties,Uses,Production
Description
Sodium acetate (CH3COONa) is the sodium salt of acetic acid. It appears as a colorless deliquescent salt with a wide range of applications. In industry, it can be used in textile industry to neutralize sulfuric acid waste streams and as a photoresist upon using aniline dyes. In concrete industry, it can be used as a concrete sealant to mitigate the water damage. In food, it can be used as a seasoning. It can also be used as a buffer solution in lab. In addition, it is also used in heating pads, hand warmers and hot ice. For laboratory use, it can be produced by the reaction between acetate with the sodium carbonate, sodium bicarbonate and sodium hydroxide. In industry, it is prepared from the glacial acetic acid and sodium hydroxide.
Chemical Properties
Sodium acetate, CH3COONa, also abbreviated NaOAc , also sodium ethanoate, is the sodium salt of acetic acid, was made by the reaction of acetic acid with sodium carbonate. It is soluble in water but less so in alcohol. This colourless salt has a wide range of uses. Sodium acetate was used as a pH modifier for toning baths.
Physical properties
Anhydrous salt is a colorless crystalline solid; density 1.528 g/cm3; melts at 324°C; very soluble in water; moderately soluble in ethanol. The colorless crystalline trihydrate has a density 1.45 g/cm3; decomposes at 58°C; is very soluble in water; pH of 0.1M aqueous solution is 8.9; moderately soluble in ethanol, 5.3 g/100mL.
Occurrence
Acetic acid or acetates are present in most plant and animal tissues in small, but detectable amounts
Uses
Used as buffers.
Acidity regulation (buffering)
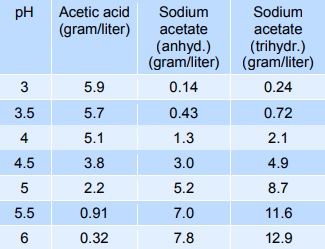
Sodium acetate mixed with acetic acid forms a pH buffer, which can be used to stabilise the pH of foods in the pH-range from 3 to 6. The table below gives indicative values of the composition needed to give a certain pH. The mixtures below can be diluted at least 10 times with minimum effect on pH, however, the stability decreases.
Synthesis
For laboratory use, sodium acetate is very inexpensive, and is usually purchased instead of being synthesized. It is sometimes produced in a laboratory experiment by the reaction of acetic acid (ethanoic acid) with sodium carbonate, sodium bicarbonate, or sodium hydroxide. These reactions produce aqueous sodium acetate and water. Carbon dioxide is produced in the reaction with sodium carbonate and bicarbonate, and it leaves the reaction vessel as a gas (unless the reaction vessel is pressurized). This is the well-known "volcano" reaction between baking soda (sodium bicarbonate) and vinegar.
CH3COOH + NaHCO3 → CH3COONa + H2O + CO2
Industrially, sodium acetate is prepared from glacial acetic acid and sodium hydroxide.
CH3COOH + NaOH → CH3COONa + H2O.
Preparation
Sodium acetate is prepared by reacting sodium hydroxide or sodium carbonate with acetic acid in aqueous solution. The solution is evaporated to obtain hydrated crystals of sodium acetate.
NaOH + CH3COOH → CH3COONa + H2O
Na2CO3 + CH3COOH → 2CH3COONa + CO2 + H2O
Definition
ChEBI: Sodium acetate is an organic sodium salt. It contains an acetate.
Reactions
Sodium acetate can be used to form an ester with an alkyl halide such as bromo ethane:
CH3COONa + Br CH2CH3→ CH3COOCH2CH3+ NaBr
Caesium salts catalyze this reaction.
General Description
Sodium Acetate is reported to inhibit the growth of Listeria monocytogenes.
Reactivity Profile
When sodium acetate reacts with strong acids, irritating, noxious vapors of acetic acid are usually produced. Sodium acetate is sufficiently basic to catalyze the violent polymerization of diketene, perhaps as well as other reactive dimers that are susceptible to polymerization in the presence of a mild base.
Biological Activity
Commonly used laboratory reagent
Safety Profile
Poison by intravenous route. Moderately toxic by ingestion. A skin and eye irritant. Migrates to food from packagmg materials. Violent reaction with F2, m03, diketene. When heated to decomposition it emits toxic fumes of Na2O.
Purification Methods
Crystallise it from acetic acid and keep it under vacuum for 10hours at 120o. Alternatively, it is crystallised from aqueous EtOH, as the trihydrate. This material can be converted to anhydrous salt by heating slowly in a porcelain, nickel or iron dish, so that the salt liquefies. Steam is evolved and the mass again solidifies. Heating is now increased so that the salt melts again. (NB: if it is heated too strongly, the salt can char; avoid this.) After several minutes, the salt is allowed to solidify and is cooled to a convenient temperature (in a desiccator) before being powdered and bottled. The water content should now be less than 0.02%. [Beilstein 2 II 113, 2 III 184, 2 IV 109.]
Sodium acetate Preparation Products And Raw materials
Raw materials
Preparation Products
1of8
chevron_right| Supplier | Tel | Country | ProdList | Advantage | Inquiry |
|---|---|---|---|---|---|
| GLR Innovations | +91 9891111994 | New Delhi, India | 4542 | 58 | Inquiry |
| SAGAR LIFE SCIENCES PVT LTD | +91-8770359041 +91-8770359041 | Gujarat, India | 81 | 58 | Inquiry |
| Vardhaman P Golechha | 9704422000 | Telangana, India | 294 | 58 | Inquiry |
| Rishi Chemical Works Pvt Ltd | +91-3322290068 +91-3322290071 | Kolkata, India | 43 | 58 | Inquiry |
| Mahadev Pharmaceuticals | +912646272307 | Gujarat, India | 26 | 58 | Inquiry |
| JAY CHEMICALS | +91-9819633090 +91-9819633090 | Maharashtra, India | 85 | 58 | Inquiry |
| S.V ENTERPRISES | +919322701159 | Mumbai, India | 150 | 58 | Inquiry |
| Cefa-Cilinas Biotics Pvt Ltd | +91-7875033155 +91-8080701561 | Maharashtra, India | 121 | 58 | Inquiry |
| JSK Chemicals | +919879767970 | Gujarat, India | 3758 | 58 | Inquiry |
| PUNJAB CHEMICALS AND CROP PROTECTION LTD | +91-7508355205 +91-7508355205 | Punjab, India | 69 | 58 | Inquiry |
| Supplier | Advantage |
|---|---|
| GLR Innovations | 58 |
| SAGAR LIFE SCIENCES PVT LTD | 58 |
| Vardhaman P Golechha | 58 |
| Rishi Chemical Works Pvt Ltd | 58 |
| Mahadev Pharmaceuticals | 58 |
| JAY CHEMICALS | 58 |
| S.V ENTERPRISES | 58 |
| Cefa-Cilinas Biotics Pvt Ltd | 58 |
| JSK Chemicals | 58 |
| PUNJAB CHEMICALS AND CROP PROTECTION LTD | 58 |
Related articles
- Effect of short-term feeding of sodium acetate on milk fat yield in dairy cows
- Supplementation with sodium acetate (NaAcet) increases milk fat production through an apparent stimulation of de novo lipogene....
- Dec 19,2023
- Sodium Acetate: Preparation, Reactions and Applications
- Sodium acetate, CH3COONa, also abbreviated NaOAc, is the sodium salt of acetic acid. This colorless deliquescent salt has a wi....
- Apr 12,2023
- Practical Applications of Sodium Acetate
- Sodium acetate salt, or simply sodium acetate, has many practical uses. It is the conjugate base of a weak acid, meaning that ....
- Nov 12,2019
127-09-3(Sodium acetate)Related Search:
1of4
chevron_right




