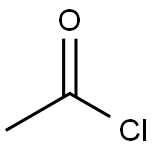
Acetyl chloride
- CAS No.
- 75-36-5
- Chemical Name:
- Acetyl chloride
- Synonyms
- AcCl;CH3COCl;ETHANOYL CHLORIDE;ACETYL-TRANS-RESVERATROL;ACETYLCLORIDE;Acetic chloride;Lidocaine Impurity 38;UN1717 (DOT);Acetylchlorid;ACETYL CHLORIDE
- CBNumber:
- CB4485487
- Molecular Formula:
- C2H3ClO
- Molecular Weight:
- 78.5
- MOL File:
- 75-36-5.mol
- MSDS File:
- SDS
- Modify Date:
- 2024/4/24 14:29:23
| Melting point | -112 °C |
|---|---|
| Boiling point | 52 °C(lit.) |
| Density | 1.104 g/mL at 25 °C(lit.) |
| vapor density | 2.7 (vs air) |
| vapor pressure | 11.69 psi ( 20 °C) |
| refractive index |
n |
| Flash point | 40 °F |
| storage temp. | Store below +15°C. |
| solubility | Miscible with acetone, chloroform, glacial acetic acid, petroleum ether, ether and benzene. |
| form | Liquid |
| Specific Gravity | approximate 1.11 |
| color | Clear colorless |
| Odor | Strong pungent odor |
| explosive limit | 7.3-19%(V) |
| Water Solubility | Decomposes violently in water |
| Sensitive | Moisture Sensitive |
| Merck | 14,85 |
| BRN | 605303 |
| Dielectric constant | 16.9(2℃) |
| Stability | Stability Highly flammable. Reacts violently with DMSO, water, lower alcohols, and amines to generate toxic fumes. May form an explosive mixture with air. Note low flash point. Incompatible with water, alcohols, amines, strong bases, strong oxidizing agents, most common metals. |
| InChIKey | WETWJCDKMRHUPV-UHFFFAOYSA-N |
| CAS DataBase Reference | 75-36-5(CAS DataBase Reference) |
| NIST Chemistry Reference | Acetyl chloride(75-36-5) |
| EPA Substance Registry System | Acetyl chloride (75-36-5) |
SAFETY
Risk and Safety Statements
| Symbol(GHS) |   GHS02,GHS05 |
|||||||||
|---|---|---|---|---|---|---|---|---|---|---|
| Signal word | Danger | |||||||||
| Hazard statements | H225-H314 | |||||||||
| Precautionary statements | P210-P233-P240-P280-P303+P361+P353-P305+P351+P338 | |||||||||
| Hazard Codes | F,C,Xn | |||||||||
| Risk Statements | 11-14-34-40-36/38 | |||||||||
| Safety Statements | 9-16-26-45-1/2-36/37 | |||||||||
| RIDADR | UN 1717 3/PG 2 | |||||||||
| WGK Germany | 1 | |||||||||
| RTECS | AO6390000 | |||||||||
| F | 3 | |||||||||
| Autoignition Temperature | 390 °C DIN 51794 | |||||||||
| TSCA | Yes | |||||||||
| HazardClass | 3 | |||||||||
| PackingGroup | II | |||||||||
| HS Code | 29159080 | |||||||||
| NFPA 704 |
|
Acetyl chloride price More Price(21)
| Manufacturer | Product number | Product description | CAS number | Packaging | Price | Updated | Buy |
|---|---|---|---|---|---|---|---|
| Sigma-Aldrich(India) | 8.22252 | Acetyl chloride for synthesis | 75-36-5 | 100ML | ₹4480 | 2022-06-14 | Buy |
| Sigma-Aldrich(India) | 8.22252 | Acetyl chloride for synthesis | 75-36-5 | 1L | ₹5320 | 2022-06-14 | Buy |
| Sigma-Aldrich(India) | 8.22252 | Acetyl chloride for synthesis | 75-36-5 | 10KG | ₹32950 | 2022-06-14 | Buy |
| Sigma-Aldrich(India) | 8.22252 | Acetyl chloride for synthesis | 75-36-5 | 25KG | ₹46479.99 | 2022-06-14 | Buy |
| Sigma-Aldrich(India) | 8.22252 | Acetyl chloride for synthesis | 75-36-5 | 8222529190 | ₹339710 | 2022-06-14 | Buy |
Acetyl chloride Chemical Properties,Uses,Production
Description
Acetic chloride is a colourless to light yellow liquid with a pungent and choking odour. Acetic chloride is highly flammable and reacts violently with DMSO, water, lower alcohols, and amines to generate toxic fumes. Along with air, acetic chloride may form an explosive mixture. It is incompatible with water, alcohols, amines, strong bases, strong oxidising agents, and most common metals. On decomposition when heated, acetic chloride produces carbon monoxide, carbon dioxide, hydrogen chloride, and phosgene.
Chemical Properties
Acetyl chloride is a highly flammable, colorless, fuming liquid with a pungent odor
Uses
Acetyl chloride acts as a reagent for the preparation of esters and amides of acetic acid. It is also useful an important reactant in Friedel-Crafts reactions as well as the introduction of an acetyl group. It serves as a starting material in the production of pharmaceutical, new plating complexing agent, acylation agent and synthetic organic intermediates.
Definition
A liquid acyl chloride used as an acetylating agent.
General Description
A colorless, fuming liquid with a pungent odor. Density 9.2 lb / gal. Flash point 40°F. Vapor, which is heavier than air, irritates the eyes and mucous membranes. Corrosive to metals and tissue.
Acetyl chloride, CH30CCI, can be prepared by treatment ofacetic acid with various reagents, such as PCl3 SOCl2 or COCI2. It can be prepared by chlorination of acetic anhydride in several different ways, by reaction of methyl chloride with carbon monoxide in the presence of catalysts, by reaction ofketene with HCI, or by partial hydrolysis of 1, 1, l-trichloroethane, Acetyl chloride hydrolyzes in the presence of water to give acetic acid. It reacts with ammonia and amines to give acetamides. Reaction with alcohols gives the corresponding acetate esters. Acetyl chloride will add across unsaturated bonds in the presence ofsuitable catalysts to give halogenated ketones.
Reactivity Profile
Acetyl chloride reacts violently with water, steam, methanol or ethanol to form hydrogen chloride and acetic acid. Reacts vigorously with bases, both organic and inorganic. Incompatible with oxidizing agents and alcohols. Produces highly toxic fumes of phosgene gas and chlorine when heated to decomposition [Sax, 9th ed., 1996, p. 35]. Reaction in a confined space with even a small amount of water may cause a violent eruption of gases [Bretherick, 5th ed., 1995, p. 281]. Vapor forms an explosive mixture with air [Kirk-Othmer, 3rd ed., Vol. 1, 1978, p. 162]. Polymerization reaction with dimethyl sulfoxide is particularly violent [Buckley, A., J. Chem. Ed., 1965, 42, p. 674]. May react vigorously or explosively if mixed with diisopropyl ether or other ethers in the presence of trace amounts of metal salts [J. Haz. Mat., 1981, 4, 291].
Health Hazard
Vapor irritates mucous membranes. Ingestion of liquid or contact with eyes or skin causes severe irritation.
Safety Profile
Poison by inhalation. Moderately toxic by ingestion. A human systemic irritant by inhalation. Violent hydrolysis reaction with water or steam produces heat, acetic acid, HCl, and other corrosive chlorides. May decompose during preparation. Dangerous fire hazard when exposed to heat or flame. Explosion hazard by spontaneous chemical reaction with dimethyl sulfoxide or ethanol. Also incompatible with Pcb. When heated to decomposition it emits highly toxic fumes of phosgene and Cl-. To fight fire, use CO2 or dry chemical. See also CHLORIDES
Potential Exposure
Acetyl chloride is used in organic synthesis as an acetylating agent and in testing for water and/ or cholesterol in organic liquids, in the pharmaceutical industry and in pesticide manufacture.
storage
Acetic chloride should be stored in a segregated and approved area, away from incompatibles such as oxidizing agents, alkalis and moisture. The container of acetic chloride should be kept in a cool, well-ventilated area, tightly sealed until ready for use. Users should avoid all possible sources of ignition, i.e., spark or flames.
Shipping
UN1717 Acetyl chloride, Hazard Class: 3; Labels: 3-Flammable liquid, 8-Corrosive material
Purification Methods
Reflux acetyl chloride with PCl5 for several hours to remove traces of acetic acid, then distil it. Redistil it from one-tenth its volume of dimethylaniline or quinoline to remove free HCl. A.R. quality is freed from HCl by pumping it for 1hour at -78o and distilling it into a trap at -196o. [Beilstein 2 IV 395.] LACHRYMATORY.
Incompatibilities
ticide manufacture. Incompatibilities: Avoid contact with moisture, steam, water, alcohols, dimethylsulfoxide, strong bases; phosphorus trichloride; oxidizers, and amines, since violent reactions may occur. Keep away from heat, fire, and welding operations.
Waste Disposal
Consult with environmental regulatory agencies for guidance on acceptable disposal practices. Generators of waste containing this contaminant (≥100 kg/mo) must conform with EPA regulations governing storage, transportation, treatment, and waste disposal. May be mixed slowly with sodium bicarbonate solution and then flushed to sewer with large volumes of water. May also be incinerated.
Acetyl chloride Preparation Products And Raw materials
Raw materials
1of2
chevron_rightPreparation Products
1of8
chevron_right| Supplier | Tel | Country | ProdList | Advantage | Inquiry |
|---|---|---|---|---|---|
| ANJI BIOSCIENCES | +91-9000100077 +91-9000100077 | Hyderabad, India | 430 | 58 | Inquiry |
| Nikava Pharmaceutical Industries | +91-9653317212 +91-9653317212 | Mumbai, India | 20 | 58 | Inquiry |
| Om Organic Chemicals | +91-9594082107 +91-9594082107 | Maharashtra, India | 5 | 58 | Inquiry |
| JSK Chemicals | +919879767970 | Gujarat, India | 3758 | 58 | Inquiry |
| Excel Industries Limited | +91-22-66464200 +91-2266464200 | Mumbai, India | 17 | 58 | Inquiry |
| Ralington Pharma | +91-7948911722 +91-9687771722 | Gujarat, India | 1350 | 58 | Inquiry |
| Veekay Chemicals | +91-8657432590 +91-9821518903 | Maharashtra, India | 37 | 58 | Inquiry |
| IOL Chemicals And Pharmaceuticals Limited | +91-9878429978 +91-9878996045 | Punjab, India | 30 | 58 | Inquiry |
| Real Chemsys Products Pvt Limited | +91-1204216880 +91-1204212000 | New Delhi, India | 29 | 58 | Inquiry |
| Sima Chemicals (Sima Products) | +91-9821136312 +91-9321375313 | Maharashtra, India | 20 | 58 | Inquiry |
Related articles
- The uses of Acetyl chloride
- Acetyl chloride is a powerful acetylating agent. It is used in the manufacture of aspirin, acetaminophen, and acetanilide. Liq....
- Apr 24,2024
- Acetyl Chloride: Preparation and Hazards
- The passage introduces the preparation and hazards of Acetyl chloride.
- Nov 22,2022
- Acetyl chloride –Chemical Properties and Application in Reagents
- Acetyl chloride [75-36-5], C2H3OCl, mol wt 78.50, is a colorless, corrosive, irritating liquid that fumes in air. It has a sti....
- Apr 1,2020
75-36-5(Acetyl chloride)Related Search:
1of4
chevron_right




