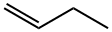1-부텐
|
|
1-부텐 속성
- 녹는점
- −185 °C(lit.)
- 끓는 점
- −6.3 °C(lit.)
- 밀도
- 0.5951
- 증기 밀도
- 1.93 (vs air)
- 증기압
- 1939 mm Hg ( 21.1 °C)
- 굴절률
- 1.3962
- 인화점
- 80
- 저장 조건
- -20°C Freezer
- 용해도
- 알코올, 벤젠, 에테르에 용해됨(Weast, 1986)
- 산도 계수 (pKa)
- >14 (Schwarzenbach et al., 1993)
- 물리적 상태
- 가스
- 색상
- 무색 내지 거의 무색
- 냄새
- 약간 향기로운
- Odor Threshold
- 0.36ppm
- 폭발한계
- 9.3%
- 수용성
- 25°C에서 222 mg/kg(진탕 플라스크-GC, McAuliffe, 1966)
- 어는점
- -185.35℃
- Merck
- 14,1519
- BRN
- 1098262
- Henry's Law Constant
- (atm?m3/mol): 0.25 at 25 °C (Hine and Mookerjee, 1975)
- 안정성
- 휘발성 물질
- InChIKey
- VXNZUUAINFGPBY-UHFFFAOYSA-N
- LogP
- 2.35 at 20℃
- CAS 데이터베이스
- 106-98-9(CAS DataBase Reference)
안전
- 위험 및 안전 성명
- 위험 및 사전주의 사항 (GHS)
| 위험품 표기 | F+,F | ||
|---|---|---|---|
| 위험 카페고리 넘버 | 12 | ||
| 안전지침서 | 9-16-33 | ||
| 유엔번호(UN No.) | UN 1012 2.1 | ||
| WGK 독일 | - | ||
| F 고인화성물질 | 4.5-31 | ||
| 자연 발화 온도 | 723 °F | ||
| 위험 참고 사항 | Extremely Flammable | ||
| DOT ClassificationII | 2.1 (Flammable gas) | ||
| 위험 등급 | 2.1 | ||
| 포장분류 | II | ||
| HS 번호 | 2901230000 | ||
| 유해 물질 데이터 | 106-98-9(Hazardous Substances Data) | ||
| 기존화학 물질 | KE-03887 |
1-부텐 C화학적 특성, 용도, 생산
물성
CMC-Na는 선상 고분자 이온 성 셀룰로오스 에테르이며 나트륨 카르복시 메틸 셀룰로오스의 알칼리 화, 에테르 화, 중화, 세척 다른 프로세스들은 반응을 일으켰다. CMC는 일반적으로 CMC라고 불리는 수용성 소금입니다. CMC는 독창적 인 점착성, 수분 흡수 및 수분 보유 성질을 가지고 있습니다. 또한, CMC는 천천히 생분해 성을 갖는 천연 유래 셀룰로오스이기 때문에 친환경적으로 만든 후 소각 처리가 가능합니다.유제 분산제
유화제는 에멀젼에서의 상간의 표면 장력을 향상시켜 균일하고 안정한 분산 시스템 또는 에멀젼을 형성하는 물질입니다. 유화제는 분자 내에 친수성기와 친수성기를 가진 표면 활성 물질입니다. 그들은 계면 장력을 감소시키고 에멀젼을 형성하는데 필요한 에너지를 감소시켜 유제의 에너지를 증가시키는 오일 / 물 계면에 집중합니다. 음이온 계면 활성제로서, 도데 실 벤젠 술폰산 나트륨은 표면 활성이 우수하고 친수성이다. 따라서, 도데 실 벤젠 술폰산 나트륨은 화장품, 식품, 인쇄 및 염색 에이즈 및 살충제와 같은 에멀젼의 제조에 널리 사용되어왔다.개요
1-Butene is a colourless, stable but polymerises exothermically, extremely flammable liquefied gas with an aromatic odour. It is insoluble in water and is one of the isomers of butane. 1-Butene readily forms explosive mixtures with air. It is incompatible with strong oxidising agents, halogens, halogen acids, metal salts, boron trifluoride, fluorine, and nitrogen oxides. 1-Butene of high purity is made by cracking naphtha and separating it from other products by an extra-high-purity distillation column. It is an important organic compound in the production of several industrial materials – for instance, linear low-density polyethylene (LLDPE), a more flexible and resilient polyethylene, and a range of polypropylene resins – and in the production of polybutene, butylene oxide, and the C4 solvents secondary butyl alcohol (SBA) and MEK. The vapour of 1-butene is heavier than air and may travel long distances to an ignition source and flash back.화학적 성질
1-Butene is a colorless, extremely flammable liquefi ed gas with an aromatic odor. It is insol uble in water and is an isomer of butane. It is highly flammable and readily forms explo sive mixtures with air. 1-Butene of high purity is made by cracking naphtha and separating it from other products by an extra-high purity distillation column. However, 1-butene is incompatible with metal salts, fl uorine, nitrogen oxides, boron trifl uoride, halogen acids, halogens, and strong oxidizing agents. It is an important organic compound in the produc tion of several industrial materials, i.e., linear low density polyethylene (LLDPE), a more fl exible and resilient polyethylene, a range of polypropylene resins, and in the production of polybutene, butylene oxide and in the C4 solvents, secondary butyl alcohol (SBA) and methyl ethyl ketone (MEK). The vapor of 1-butene is heavier than air and may travel long distances to an ignition source and fl ash back.물리적 성질
Butenes or butylenes are hydrocarbon alkenes that exist as four different isomers. Each isomer is a flammable gas at normal room temperature and one atmosphere pressure, but their boiling points indicate that butenes can be condensed at low ambient temperatures and/or increase pressure similar to propane and butane. The “2” designation in the names indicates the position of the double bond. The cis and trans labels indicate geometric isomerism. Geometric isomers are molecules that have similar atoms and bonds but different spatial arrangement of atoms. The structures indicate that three of the butenes are normal butenes, n-butenes, but that methylpropene is branched. Methylpropene is also called isobutene or isobutylene. Isobutenes are more reactive than n-butenes, and reaction mechanisms involving isobutenes differ from those of normal butenes.용도
Butenes are used extensively in gasoline production to produce high-octane gasoline compounds.Another large use of normal butenes in the petrochemical industry is in the production of 1,3-butadiene. Butene is used in the plastics industry to make both homopolymers and copolymers. Another use of 1-butene is in the production of solvents containing four carbons such as secondary butyl alcohol and methyl ethyl ketone (MEK).생산 방법
Most butenes are produced in the cracking process in refineries along with other C-4 fractions such as the butanes. Butenes are separated from other compounds and each other by several methods. Isobutene is separated from normal butanes by absorption in a sulfuric acid solution. Normal butenes can be separated from butanes by fractionation. The close boiling points of butanes and butenes make straight fractional distillation an inadequate separation method, but extractive distillation can be used. Butenes can also be prepared from the dehydrogenation (elimination of hydrogen) of butane.정의
ChEBI: A butene with unsaturation at position 1.일반 설명
Colorless gas.공기와 물의 반응
Highly flammable. Insoluble in water.반응 프로필
The unsaturated aliphatic hydrocarbons, such as 1-BUTENE, are generally much more reactive than the alkanes. Strong oxidizers may react vigorously with them. Reducing agents can react exothermically to release gaseous hydrogen. In the presence of various catalysts (such as acids) or initiators, compounds in this class can undergo very exothermic addition polymerization reactions. May react with oxidizing materials. Aluminum borohydride reacts with alkenes and in the presence of oxygen, combustion is initiated even in the absence of moisture.건강위험
Exposures to 1-butene cause the effects of an asphyxiant and/or an anesthetic (at high concentrations). Workers exposed to 1-butene develop eye irritation.화재위험
1-BUTENE is flammable. Vapor is heavier than air and may travel long distances to an ignition source and flash back.환경귀착
Biological. Biooxidation of 1-butene may occur yielding 3-buten-1-ol, which may oxidize to give 3-butenoic acid (Dugan, 1972). Washed cell suspensions of bacteria belonging to the genera Mycobacterium, Nocardia, Xanthobacter, and Pseudomonas and growing on selected alkenes metabolized 1-butene to 1,2-epoxybutane (Van Ginkel et al., 1987).Photolytic. Products identified from the photoirradiation of 1-butene with nitrogen dioxide in air are epoxybutane, 2-butanone, propanal, ethanol, ethyl nitrate, carbon monoxide, carbon dioxide, methanol, and nitric acid (Takeuchi et al., 1983).
The following rate constants were reported for the reaction of 1-butene and OH radicals in the atmosphere: 1.0 x 10-17 cm3/molecule?sec (Bufalini and Altshuller, 1965); 2.70 x 10-11 cm3/molecule?sec (Atkinson et al., 1979); 3.14 x 10-11 cm3/molecule?sec (Atkinson, 1990; Sablji? and Güsten, 1990). Reported photooxidation reaction rate constants for the reaction of 1-butene and ozone are 1.23 x 10-17, 1.0 x 10-17, 1.03 x 10-17 cm3/molecule?sec (Adeniji et al., 1981). Based on the reaction of 1-butene and OH radicals gas phase, the atmospheric lifetime was estimated to be 5.5 h in summer sunlight.
Chemical/Physical. Complete combustion in air yields carbon dioxide and water. Incomplete combustion will generate carbon monoxide.
Hydrolysis in water is not expected to be signicant because 1-butene is very volatile.
주의 사항
When working with 1-butene, occupational workers should wear proper protectives, preferably a NIOSH-approved full-face positive pressure supplied-air respirator or a self contained breathing apparatus (SCBA). Workers should not wear contact lenses.1-부텐 준비 용품 및 원자재
원자재
sec-부탄올
1,3-부타디엔
이소부탄올
아이소부탄
고무시멘트 희석제
부탄
n-부탄올
1-(2-메틸시클로프로필)에타논
n-트리데칸
헥센(시스-2)
P-TOLUENESULFONICACIDSEC-BUTYL에스테르
2-메틸헥산
2-메틸옥탄
2-플루오로부탄
노르말노난
2-METHYLDECANE
PROPARGYLALDEHYDE DIETHYL ACETAL
시스-2-부텐
준비 용품
1-부텐 공급 업체
글로벌( 160)공급 업체
| 공급자 | 전화 | 이메일 | 국가 | 제품 수 | 이점 |
|---|---|---|---|---|---|
| Henan Tianfu Chemical Co.,Ltd. | +86-0371-55170693 +86-19937530512 |
info@tianfuchem.com | China | 21666 | 55 |
| career henan chemical co | +86-0371-86658258 +8613203830695 |
sales@coreychem.com | China | 29888 | 58 |
| Xi'an Kono chem co., Ltd., | 029-86107037 13289246953 |
info@konochemical.com | China | 2995 | 58 |
| Hubei xin bonus chemical co. LTD | 86-13657291602 |
linda@hubeijusheng.com | CHINA | 22968 | 58 |
| Chongqing Chemdad Co., Ltd | +86-023-6139-8061 +86-86-13650506873 |
sales@chemdad.com | China | 39916 | 58 |
| Hefei TNJ Chemical Industry Co.,Ltd. | +86-0551-65418671 +8618949823763 |
sales@tnjchem.com | China | 34571 | 58 |
| Shaanxi Didu New Materials Co. Ltd | +86-89586680 +86-13289823923 |
1026@dideu.com | China | 8672 | 58 |
| Shanghai Acmec Biochemical Technology Co., Ltd. | +undefined18621343501 |
product@acmec-e.com | China | 33350 | 58 |
| SHANGHAI KEAN TECHNOLOGY CO., LTD. | +8613817748580 |
cooperation@kean-chem.com | China | 40068 | 58 |
| Mainchem Co., Ltd. | +86-0592-6210733 |
sale@mainchem.com | China | 32360 | 55 |
1-부텐 관련 검색:
아세틸아세톤산 구리 비스(2-프로판올에이토)비스(2,4-펜탄디오네이토)티타늄 옥소비스(펜탄-2,4-디오나토-O,O"")바나듐 아세틸아세토산 제2철 디스프로슘 트리(2,2,6,6-테트라메틸-3,5-헵탄디오네이트) 루테늄(III)아세틸아세토네이트 니켈 아세틸아세톤산 트리(2,2,6,6-테트라메틸-3,5-헵타네디오나토)이테르븀 알루미늄 아세틸아세토네이트 프라세오디뮴 트리(디피발로일메탄) 실버 아세틸아세토네이트 코발트(II) 아세틸아세토네이트 하이드레이트 바나듐(III) 아세틸아세토네이트 2-메틸-2-부텐 아밀렌 부틸렌옥사이드 트랜스-2-부텐 크로틸알콜