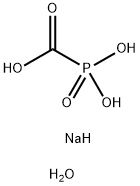
Phosphonoformic acid trisodium salt hexahydrate
|
|
Phosphonoformic acid trisodium salt hexahydrate 속성
- 저장 조건
- 2-8°C
- 용해도
- H2O: 0.1 g/mL 뜨겁고 투명하며 무색입니다.
- 수용성
- 물에 용해됩니다.
- InChIKey
- ILRVASBWNRYBFD-UHFFFAOYSA-K
- CAS 데이터베이스
- 34156-56-4(CAS DataBase Reference)
안전
- 위험 및 안전 성명
- 위험 및 사전주의 사항 (GHS)
Phosphonoformic acid trisodium salt hexahydrate C화학적 특성, 용도, 생산
화학적 성질
Phosphonoformic acid trisodium salt hexahydrate is white or almost white, crystalline powder.정의
ChEBI: The hexahydrate form of trisodium phosphonoformate. It is used as an antiviral agent in the treatment of cytomegalovirus retinitis (CMV retinitis, an inflamation of the retina that can lead to blindness) and as an alternative to ganciclovir for AIDS patien s who require concurrent antiretroviral therapy but are unable to tolerate ganciclovir due to haematological toxicity.원료
Phosphonoformic acid trisodium salt hexahydrate can be generated in vitro, and CMV strains resistant to both ganciclovir and foscarnet have occasionally been recovered from humans.Pharmaceutical Applications
Phosphonoformic acid trisodium salt hexahydrate is a synthetic non-nucleoside pyrophosphate analog formulated as the trisodium hexahydrate for intravenous use. The solubility in water at pH 7 is only about 5% (w/w).Pharmacokinetics
Oral absorption: c. 17%Cmax 60 mg/kg intravenous 8-hourly: 557 μmol/L
Plasma half-life: 3.3–6.8 h
Volume of distribution: 0.52–0.74 L/kg
Plasma protein binding: 14–17%
Absorption and distribution
Oral bioavailability is poor. A wide range of plasma concentrations was noted (75–500 μmol/L) during 3–21 days of continuous intravenous infusion of 0.14–0.19 mg/kg per min. During continuous intravenous therapy the concentrations reached a plateau on day 3. Considerable differences in steady-state plasma concentrations exist between individuals. Drug penetrates the CSF; the mean concentration is about 40–60% of the mean plasma concentration, depending upon dose.
Metabolism and excretion
Elimination appears to be triphasic, with two initially short half-lives of 0.5–1.4 h and 3.3–6.8 h, followed by a long terminal phase of 88 h. About 88% of the cumulative intravenous dose is recovered unchanged in the urine within a week of stopping an infusion, indicating that the drug is not significantly metabolized. Non-renal clearance accounts for 14–18% of total clearance and may relate to uptake into bone. Plasma clearance decreases markedly with decreased renal function and the elimination half-life may be increased by up to 10-fold. Conventional dialysis eliminates about 25% of a dose while high-flux dialysis can remove nearly 60%.
Clinical Use
Treatment of CMV retinitis in patients for whom ganciclovir is contraindicated, inappropriate or ineffectiveIt is also potentially of value in the treatment of aciclovir-resistant HSV infection.
부작용
Treatment is more frequently limited by toxicity than with ganciclovir. Renal toxicity is most common. A two- to three-fold increase in serum creatinine levels occurs in 20–60% (mean 45%) of patients given 130–230 mg/kg per day as a continuous intravenous infusion. Renal impairment usually develops within the first few weeks of treatment and is generally reversible within several weeks of discontinuing therapy. Foscarnet chelates metal ions, and serum electrolyte abnormalities – predominantly hypocalcemia, hypomagnesemia, hypokalemia and hypophosphatemia – occur in about 30, 15, 16 and 8% of patients, respectively. Convulsions occur in 10–15%. Other side effects include anemia (25–50%), penile or vulval ulceration (3–9%), nausea and vomiting (20–30%), local irritation and thrombophlebitis at the infusion site, abdominal pain and occasional pancreatitis, headache (c. 25%), dizziness, involuntary muscle contractions, tremor, hypoesthesia, ataxia, neuropathy, anxiety, nervousness, depression and confusion, and skin rash. Nephrogenic diabetes insipidus has been reported.Foscarnet is contraindicated in pregnancy. Topical application does not result in dermal toxicity similar to that produced by phosphonacetic acid.
Phosphonoformic acid trisodium salt hexahydrate 준비 용품 및 원자재
원자재
준비 용품
Phosphonoformic acid trisodium salt hexahydrate 공급 업체
글로벌( 163)공급 업체
| 공급자 | 전화 | 이메일 | 국가 | 제품 수 | 이점 |
|---|---|---|---|---|---|
| Henan Fengda Chemical Co., Ltd | +86-371-86557731 +86-13613820652 |
info@fdachem.com | China | 7845 | 58 |
| ATK CHEMICAL COMPANY LIMITED | +undefined-21-51877795 |
ivan@atkchemical.com | China | 32480 | 60 |
| career henan chemical co | +86-0371-86658258 |
sales@coreychem.com | China | 29914 | 58 |
| Wuhan Chemwonders Technology Inc. | 027-85778276 |
info@chemwonders.com | CHINA | 176 | 58 |
| Xiamen AmoyChem Co., Ltd | +86-592-6051114 +8618959220845 |
sales@amoychem.com | China | 6387 | 58 |
| Hubei xin bonus chemical co. LTD | 86-13657291602 |
linda@hubeijusheng.com | CHINA | 22968 | 58 |
| Chongqing Chemdad Co., Ltd | +86-023-61398051 +8613650506873 |
sales@chemdad.com | China | 39916 | 58 |
| CONIER CHEM AND PHARMA LIMITED | +8618523575427 |
sales@conier.com | China | 49390 | 58 |
| Shaanxi Dideu Medichem Co. Ltd | 18192627656 |
1012@dideu.com | China | 3422 | 58 |
| AFINE CHEMICALS LIMITED | 0571-85134551 |
info@afinechem.com | CHINA | 15377 | 58 |
Phosphonoformic acid trisodium salt hexahydrate 관련 검색:
포름산나트륨
FOSCARNET SODIUM, DELIVERED >= 97% PURE WITH HPLC UV CHROMATOGRAM
FOSCARNET = DIHYDROXYPHOSPHINECARBOXYLIC ACID OXIDE TRISODIUM SALT
FOSCARNET IMPURITY BDISODIUM(ETHOXYOXYDOPHOSPHANYL)FORMATE EPF(CRM STANDARD)
FOSCARNET SODIUM BP STANDARD(CRM STANDARD)
FOSCARNET SODIUM ASSAY STANDARD BP(CRM STANDARD)
FOSCARNET SODIUM HEXAHYDRATE
FOSCARNET IMPURITY B
Foscarnet sodium
FOSCARNET SODIUM HEXAHYDRATE EPF(CRM STANDARD)
Phosphonoformic acid trisodium salt hexahydrate
Foscarnet
PhosphonoMethanol
FOSCARNET SODIUM, [14C]-