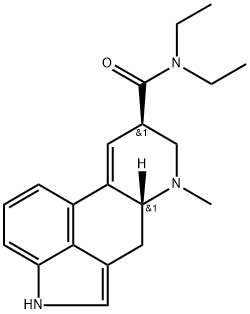
리세르그산 디에틸아마이드
|
|
리세르그산 디에틸아마이드 속성
- 녹는점
- 91-93?C
- 알파
- D20 +17° (c = 0.5 in pyridine)
- 끓는 점
- 461.9°C (rough estimate)
- 밀도
- 1.1021 (rough estimate)
- 굴절률
- 1.6200 (estimate)
- 인화점
- 2℃
- 저장 조건
- −20°C
- 용해도
- 클로로포름(약간 용해됨), DMSO(약간 용해됨), 메탄올(약간 용해됨, 가열)
- 물리적 상태
- 고체
- 물리적 상태
- 단단한 모양
- 산도 계수 (pKa)
- pKa 7.5 (Uncertain)
- 색상
- 옅은 갈색에서 갈색까지
- 안정성
- 안정적인. 강한 산화제와 호환되지 않습니다.
안전
- 위험 및 안전 성명
- 위험 및 사전주의 사항 (GHS)
| 위험품 표기 | T+,T,F,Xn | ||
|---|---|---|---|
| 위험 카페고리 넘버 | 26/27/28-40-36/38-23/25-36-20/21/22-11 | ||
| 안전지침서 | 22-28-36-45-33-24-16-7-36/37-26 | ||
| 유엔번호(UN No.) | UN 2811 6.1/PG 1 | ||
| WGK 독일 | 3 | ||
| RTECS 번호 | KE4100000 | ||
| 유해 물질 데이터 | 50-37-3(Hazardous Substances Data) | ||
| 독성 | LD50 in mice, rats, rabbits (mg/kg): 46, 16.5, 0.3 i.v. | ||
| DEA Controlled Substances | CSCN: 7310 CAS SCH: III NARC: N |
||
| DEA Controlled Substances | CSCN: 7310 CAS SCH: III NARC: N |
리세르그산 디에틸아마이드 C화학적 특성, 용도, 생산
개요
Lysergic acid diethylamide (LSD) is synthetically derived from the fungus, Claviceps purpuria. Several isomers of this compound exist, although only the D-isomer is considered active. LSD was first synthesized by Albert Hofmann in 1938 in a search for an analeptic drug. After finding its psychogenic activity, it was marketed by Sandoz laboratories under the name Delysid?. It was also reportedly used by the Central Intelligence Agency as a tool for interrogation. Due to its potential for ‘bad trips,’ ‘flashbacks,’ and potential risk for brain injury, LSD was banned federally in 1966 and is currently only used illicitly. LSD is chemically similar to other naturally occurring lysergamides found in multiple species of morning glory.화학적 성질
crystalline solid용도
LSD is obtained by partial synthesis fromD-lysergic acid. It is also produced bymicrobial reaction of Claviceps paspali overthe hydroxylethylamide. It is a well-knownhallucinogen and a drug of abuse, listed asa controlled substance in the U.S. Code ofFederal Regulations (Title 21, Part 1308.11,1987). It is used as an antagonist to serotoninand in the study and treatment of mentaldisorders.정의
ChEBI: An ergoline alkaloid arising from formal condensation of lysergic acid with diethylamine.일반 설명
Prismatic crystals (from benzene). Tasteless and odorless. A hallucinogen.반응 프로필
An amide. Organic amides/imides react with azo and diazo compounds to generate toxic gases. Flammable gases are formed by the reaction of organic amides/imides with strong reducing agents. Amides are very weak bases (weaker than water). Imides are less basic yet and in fact react with strong bases to form salts. That is, they can react as acids. Mixing amides with dehydrating agents such as P2O5 or SOCl2 generates the corresponding nitrile. The combustion of these compounds generates mixed oxides of nitrogen (NOx).건강위험
LSD is a strong psychedelic agent. Theeffects in human are excitement, euphoria,hallucinations, and distorted perceptions.It alters the thinking process, producingillusions and loss of contact with reality.In humans, a dose (intramuscular)of 0.7–0.9 mg/kg or an oral dose of2.5–3.0 mg/kg may produce the effectsabove. Other symptoms may include nausea,vomiting, dilation of pupils, restlessness, andperipheral vascoconstriction. However, thereis no reported case of overdose death. In rabbits,somnolence, ataxia, and an increase inbody temperature were the symptoms notedat the LD50 (intravenous) doses at 0.3 mg/kg.LD50 value, intravenous (mice): 46 mg/kg
LD50 value, subcutaneous (guinea pigs):16 mg/kg.
화재위험
Flash point data for LSD are not available; however LSD is probably combustible.Safety Profile
Poison by ingestion, subcutaneous, intraperitoneal, and intravenous routes. Mutation data reported. Human systemic effects by ingestion and intramuscular routes: euphoria, hallucinations, distorted perceptions, excitement, anorexia, nausea and vomiting. An experimental teratogen. Other experimental reproductive effects. Mutation data reported. A much-abused hallucinogen. A federally regulated substance. When heated to decomposition it emits toxic fumes of NOx환경귀착
LSD may exist in the air or soil. In the air, LSD may be susceptible to photochemical reactions with subsequently induced radical analogs. Photolysis and oxidative degradation may occur with an airborne half-life of 18 min. LSD’s pKa of 7.8 will have a meaningful percentage of the drug in the cationic form allowing it to interact with soil.리세르그산 디에틸아마이드 준비 용품 및 원자재
원자재
준비 용품
리세르그산 디에틸아마이드 관련 검색:
다이에틸아민 에틸렌디아민 리세르그산 디에틸아마이드
LSD-D3
Ergoline-8-beta-carboxamide, 9,10-didehydro-N,N-diethyl-6-methyl-, tar trate (1:1), d-
LSD REAGENT
ANTI-LSD1 (AOF2) (N-TERMINAL)
ISO-LSD
Bromo-LSD
LSD-25-Pyrrolidate
NOR-LSD AND NOR-ISO-LSD
(+)-LSD tartrate,D-LSD tartrate,LSD 25 tartrate,LSD tartarate
LSD maleate
LSD hydrobromide
Daito Black Base LSD.
2-OXO-3-HYDROXY-LSD
N1-Methyl-2-iodo-125I-LSD
SIGMA PSEUDO(TM) NARCOTIC SCENT LSD FORMULATION