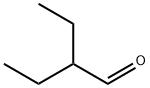
2 에틸부틸알데히드 C화학적 특성, 용도, 생산
화학적 성질
2-Ethylbutyraldehyde has a pungent odor.
출처
Reported found in melon, French fried potato, wheaten bread, scallops, citrus fruits, white bread and maize.
용도
2-Ethylbutyraldehyde has been used in the preparation of aldoximes using aqueous hydroxylamine.
일반 설명
A clear colorless liquid. Flash point 70°F. Less dense than water and insoluble in water. Vapors heavier than air.
공기와 물의 반응
Highly flammable. With air slowly form peroxides. Insoluble in water.
반응 프로필
2-ETHYLBUTYRALDEHYDE is an aldehyde. Aldehydes are frequently involved in self-condensation or polymerization reactions. These reactions are exothermic; they are often catalyzed by acid. Aldehydes are readily oxidized to give carboxylic acids. Flammable and/or toxic gases are generated by the combination of aldehydes with azo, diazo compounds, dithiocarbamates, nitrides, and strong reducing agents. Aldehydes can react with air to give first peroxo acids, and ultimately carboxylic acids. These autoxidation reactions are activated by light, catalyzed by salts of transition metals, and are autocatalytic (catalyzed by the products of the reaction). The addition of stabilizers (antioxidants) to shipments of aldehydes retards autoxidation.
위험도
Irritant to eyes and skin. Flammable, dangerous fire risk.
건강위험
May cause toxic effects if inhaled or absorbed through skin. Inhalation or contact with material may irritate or burn skin and eyes. Fire will produce irritating, corrosive and/or toxic gases. Vapors may cause dizziness or suffocation. Runoff from fire control or dilution water may cause pollution.
화재위험
HIGHLY FLAMMABLE: Will be easily ignited by heat, sparks or flames. Vapors may form explosive mixtures with air. Vapors may travel to source of ignition and flash back. Most vapors are heavier than air. They will spread along ground and collect in low or confined areas (sewers, basements, tanks). Vapor explosion hazard indoors, outdoors or in sewers. Runoff to sewer may create fire or explosion hazard. Containers may explode when heated. Many liquids are lighter than water.
Safety Profile
Moderately toxic by
ingestion. Mildly toxic by inhalation. A skin
irritant. Flammable liquid. Can react
vigorously with oxidizing materials. To fight
fire, use alcohol foam, Co2, dry chemical.
When heated to decomposition it emits
acrid smoke and fumes. See also
ALDEHYDES.
잠재적 노출
Used in organic synthesis of pharmaceuticals and rubber chemicals.
운송 방법
UN1178 2-Ethyl butyraldehyde, Hazard Class: 3;
Labels: 3-Flammable liquid
비 호환성
Vapors may form explosive mixture with
air. Incompatible with oxidizers (chlorates, nitrates, peroxides, permanganates, perchlorates, chlorine, bromine, fluorine, etc.); contact may cause fires or explosions. Keep
away from alkaline materials, strong bases, strong acids,
oxoacids, epoxides, and reducing agents. Aldehydes are frequently involved in self-condensation or polymerization
reactions. These reactions are exothermic; they are often
catalyzed by acid. Aldehydes are readily oxidized to give
carboxylic acids. Flammable and/or toxic gases are generated by the combination of aldehydes with azo, diazo compounds, dithiocarbamates, nitrides, and strong reducing
agents. Aldehydes can react with air to give first peroxo
acids, and ultimately carboxylic acids. These autoxidation
reactions are activated by light, catalyzed by salts of transition metals, and are autocatalytic (catalyzed by the products
of the reaction). The addition of stabilizers (antioxidants) to
shipments of aldehydes retards autoxidation
폐기물 처리
Dissolve or mix the material
with a combustible solvent and burn in a chemical incinerator equipped with an afterburner and scrubber. All federal,
state, and local environmental regulations must be
observed. Consult with environmental regulatory agencies
for guidance on acceptable disposal practices. Generators
of waste containing this contaminant (≥100 kg/mo) must
conform with EPA regulations governing storage, transportation, treatment, and waste disposal.
2 에틸부틸알데히드 준비 용품 및 원자재
원자재
준비 용품