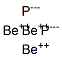beryllium phosphide
|
|
beryllium phosphide 속성
안전
beryllium phosphide C화학적 특성, 용도, 생산
개요
Beryllium phosphide has the formula of Be3P2 and the molecular weight of 88.9841 g/mol. It can be prepared by reaction of the elements, but not from aqueous solution:12Be + 2P4 ? 4Be3P2
Red phosphorus melts at 317.3 C and the boiling point is 553°C so that addition of Be metal serves to form the phosphide. By adding Be to P4 at about 350°C, but less than the phosphorus sublimation temperature of 415°C in an inert atmosphere, the melt readily forms the phosphide. A better way is to sublime the P4 at 450°C in an inert gas stream and react it with Be metal at 750°C. Be3P2 has the CAS number 58127-61-0. It is a brown powder having a slight odor of garlic. Once formed, Be3P2 is very stable. However, it reacts slowly with moisture to form phosphine, PH3 (which is toxic and flammable), and must be stored under dry conditions.
용도
Beryllium phosphide is a semiconductor and has been studied by XRD methods using powder or a crystal, and by neutron diffraction from powder. example, is 160% wider. It is for this reason that beryllium phosphide films or composites have found little usage in the industry.beryllium phosphide 준비 용품 및 원자재
원자재
준비 용품
beryllium phosphide 공급 업체
글로벌( 0)공급 업체
| 공급자 | 전화 | 이메일 | 국가 | 제품 수 | 이점 |
|---|
beryllium phosphide 관련 검색:
인화 칼슘 베릴륨
Magnesium phosphide
Calcium hypophosphite
Magnesium hypophosphite
strontium phosphide
CALCIUM PHOSPHITE
BARIUM PHOSPHITE
Radium Phosphide
Strontium Hypophosphite
Radium Hypophosphite
Beryllium Hypophosphite
MAGNESIUM PHOSPHITE
BARIUM PHOSPHIDE
Radium Phosphite
Strontium Phosphite
BARIUM HYPOPHOSPHITE
Beryllium Phosphite