|
ChemicalBook Optimization Suppliers |
|
| 化学名: | ジフェニルジフルオロシラン | | 英語化学名: | DIPHENYLDIFLUOROSILANE | | 别名: | difluorodiphenyl-silan;Diphenylsilane difluoride;DIPHENYLDIFLUOROSILANE;Diphenyl difluorosilanegas cylinder;DIFLUORODIPHENYLSILANE;Difluorodiphenylsilane97%;Benzene,1,1'-(difluorosilylene)bis-;ifluoro(diphenyl)silane | | CAS番号: | 312-40-3 | | 分子式: | C12H10F2Si | | 分子量: | 220.29 | | EINECS: | 206-226-8 | | カテゴリ情報: | | | Mol File: | 312-40-3.mol | 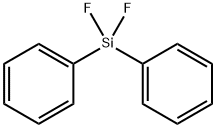 |
| 主な危険性 | C | | Rフレーズ | 34 | | Sフレーズ | 26-36/37/39-45 | | RIDADR | 1760 | | Hazard Note | Corrosive | | TSCA | Yes | | HSコード | 2931900090 |
| | ジフェニルジフルオロシラン Usage And Synthesis |
| 物理的性質 | bp 246–247 °C, bp 156°C50 mmHg, bp
66–70°C0.5 mmHg; d 1.155 g cm?1. | | 使用 | A moisture insensitive equivalent of diphenyldichlorosilane that is
more reactive toward nucleophiles. Source of phenyl group in the
Hiyama coupling reaction. More stable to electrophilic aromatic
substitution than diphenyldichlorosilane.
Eaborn reported that fluorosilanes are more reactive toward organometallic reagents than the
corresponding chlorosilanes, and therefore more suited for prepa�ration of sterically hindered organosilanes. | | 製造方法 | commercially available. Readily prepared
by oxidation of diphenylsilane with CuF2, or by treatment
of diphenyldichlorosilane with HF, ZnF2, NaBF4, or
(NH4)2SiF6. Rate enhancements of the chlorine–fluorine exchange have been achieved with ultrasound and addition
of water. Diethoxydiphenylsilane and related structures are
converted to diphenyldifluorosilane with 48% HF(aq). |
|