|
ChemicalBook Optimization Suppliers |
| 名前: |
Alfa Aesar |
| 電話番号: |
400-6106006 |
| 電子メール: |
saleschina@alfa-asia.com |
| 名前: |
Energy Chemical |
| 電話番号: |
021-021-58432009 400-005-6266 |
| 電子メール: |
sales8178@energy-chemical.com |
| 名前: |
BOC Sciences |
| 電話番号: |
1-631-485-4226; 16314854226 |
| 電子メール: |
info@bocsci.com |
|
| | 2,4-ジヒドロキシ-6-メチル安息香酸 製品概要 |
| 化学名: | 2,4-ジヒドロキシ-6-メチル安息香酸 | | 英語化学名: | 2,4-DIHYDROXY-6-METHYLBENZOIC ACID | | 别名: | 2,4-DIHYDROXY-6-METHYLBENZOIC ACID;ORSELLINIC ACID;ORSELLINIC ACID MONOHYDRATE;2,4-Dihydroxy-6-methylbenzoic acid hydrate, 97%;2-Methyl-4,6-dihydroxybenzoic acid;6-Methyl-2,4-dihydroxybenzoic acid;Orsellic acid;o-orsellinic acid | | CAS番号: | 480-64-8 | | 分子式: | C8H8O4 | | 分子量: | 168.15 | | EINECS: | | | カテゴリ情報: | Inhibitors;Aromatic Carboxylic Acids, Amides, Anilides, Anhydrides & Salts | | Mol File: | 480-64-8.mol | 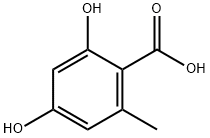 |
| | 2,4-ジヒドロキシ-6-メチル安息香酸 物理性質 |
| 融点 | 173-174°C | | 沸点 | 257.07°C (rough estimate) | | 比重(密度) | 1.3037 (rough estimate) | | 屈折率 | 1.5090 (estimate) | | 貯蔵温度 | Sealed in dry,Room Temperature | | 溶解性 | Ethyl Acetate (Slightly), Methanol (Slightly) | | 外見 | Solid | | 酸解離定数(Pka) | pK (25°) 3.90 | | 色 | White to Pale Gray | | 水溶解度 | Soluble in water, dimethyl sulfoxide or 100% ethanol. | | 安定性: | Hygroscopic | | CAS データベース | 480-64-8 |
| | 2,4-ジヒドロキシ-6-メチル安息香酸 Usage And Synthesis |
| 外観 | 白色~うすい灰褐色、結晶性粉末~粉末 | | 説明 | Orsellinic acid is a fungal metabolite and benzoic acid derivative with antioxidant and neuroprotective activities. It scavenges 2,2-diphenyl-1-picrylhydrazyl (DPPH; ) radicals with an IC50 value of 5 mM. Orsellinic acid (1 μg/ml) prevents PARP cleavage induced by platelet-activating factor (PAF) in PC12-AC cells and PAF-induced cytotoxicity in PAF receptor null (Pafr-/-) mouse cerebellar granule cells. | | 使用 | Orsellinic acid is a benzoic acid compound shown to block PAF-mediated neuronal apoptosis. | | 定義 | ChEBI: A dihydroxybenzoic acid that is 2,4-dihydroxybenzoic acid in which the hydrogen at position 6 is replaced by a methyl group. | | 生合成 | One of the simplest polyketide derivatives, orsellinic acid is widely distributed in fungi. It is biosynthesised from an acetate starter unit and three malonyl units, as shown in Figure. Intramolecular condensation of the polyketide chain gives orsellinic acid without further modification.
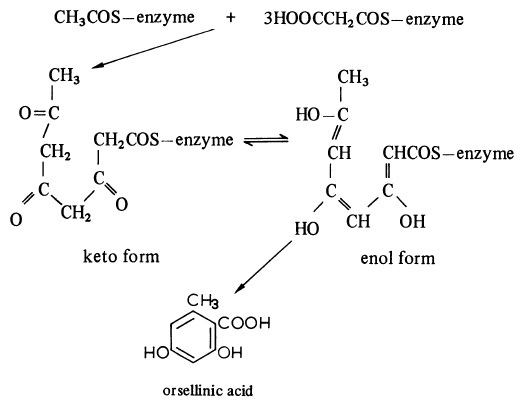
The biosynthesis of orsellinic acid. | | Synthesis Reference(s) | The Journal of Organic Chemistry, 30, p. 3566, 1965 DOI: 10.1021/jo01021a507 |
|