|
|
| | Glyoxal bis(2,6-diisopropylanil), N,Nμ-Bis(2,6-diisopropylphenyl)-1,4-diazabutadiene, N,Nμ-Bis(2,6-diisopropylphenyl)ethanediimine Basic information |
| Product Name: | Glyoxal bis(2,6-diisopropylanil), N,Nμ-Bis(2,6-diisopropylphenyl)-1,4-diazabutadiene, N,Nμ-Bis(2,6-diisopropylphenyl)ethanediimine | | Synonyms: | N,N'-1,2-Ethanediylidenebis[2,6-bis(1-methylethyl)phenylamine];N,N'-Bis(2,6-diisopropylphenyl)-1,4-diazabutadiene;N,N'-Bis(2,6-diisopropylphenyl)ethanediimine;N,N'-Bis(2,6-diisopropylphenyl)glyoxaldiimine;1,4-Bis(2,6-diisopropylphenyl)-1,4-diaza-1,3-butadiene;N,N'-(1,2-Ethanediylidene)bis(2,6-diisopropylaniline);N,N'-(1,2-Ethanediylidene)bis[2,6-bis(1-methylethyl)benzenamine];N,N'-(Ethane-1,2-diylidene)bis(2,6-diisopropylaniline) | | CAS: | 74663-75-5 | | MF: | C26H36N2 | | MW: | 376.58 | | EINECS: | | | Product Categories: | | | Mol File: | 74663-75-5.mol | 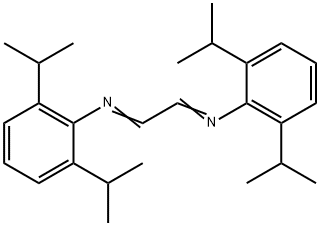 |
| | Glyoxal bis(2,6-diisopropylanil), N,Nμ-Bis(2,6-diisopropylphenyl)-1,4-diazabutadiene, N,Nμ-Bis(2,6-diisopropylphenyl)ethanediimine Chemical Properties |
| Melting point | 105-109 °C | | Boiling point | 492.0±55.0 °C(Predicted) | | density | 0.95 | | pka | 1.85±0.50(Predicted) |
| | Glyoxal bis(2,6-diisopropylanil), N,Nμ-Bis(2,6-diisopropylphenyl)-1,4-diazabutadiene, N,Nμ-Bis(2,6-diisopropylphenyl)ethanediimine Usage And Synthesis |
| Uses | Reactant in preparation of derived ruthenium olefin metathesis catalysts 1 N-cyclic carbene ligand 2 Catalyst in: Palladium-catalyzed aerobic alc. oxidn. supported by a-diimine ligands 3 Regioselective alkylation in presence of ruthenium-bisimine catalytic precursors 4 N-arylation of aromatic amines 5 Preparation of ruthenium nitrosyl alpha-diimine and iminoketone complexes as catalysts for transfer hydrogenation of ketones and atom transfer radical polymerization reactions. | | Uses | - Reactant in preparation of derived ruthenium olefin metathesis catalysts
- N-cyclic carbene ligand
Catalyst in:
- Palladium-catalyzed aerobic alc. oxidn. supported by a-diimine ligands
- Regioselective alkylation in presence of ruthenium-bisimine catalytic precursors
- N-arylation of aromatic amines
- Preparation of ruthenium nitrosyl alpha-diimine and iminoketone complexes as catalysts for transfer hydrogenation of ketones and atom transfer radical polymerization reactions
|
| | Glyoxal bis(2,6-diisopropylanil), N,Nμ-Bis(2,6-diisopropylphenyl)-1,4-diazabutadiene, N,Nμ-Bis(2,6-diisopropylphenyl)ethanediimine Preparation Products And Raw materials |
|