| Company Name: |
GLP Pharma Standards
|
| Tel: |
+91 9866074638 |
| Email: |
info@glppharmastandards.com |
| Products Intro: |
Product Name:Solifenacin EP Impurity-D
CAS:2216750-52-4
Purity:98% Package:50 MG; 100 MG; 250 MG ; 250 MG
|
| Company Name: |
S.Z. PhyStandard Bio-Tech. Co., Ltd.
|
| Tel: |
0755-4000505016 13380397412 |
| Email: |
3001272453@qq.com |
| Products Intro: |
Product Name:Solifenacin Impurity D
CAS:2216750-52-4
Purity:99% HPLC Package:10mg;25mg;50mg;100mg
|
| Company Name: |
Beijing Comparison Pharmaceutical Technology Co., Ltd
|
| Tel: |
010-52878169 15701683040 |
| Email: |
sales@bjcomparison.com |
| Products Intro: |
Product Name:[(1R)-1-phenyl-3,4-dihydroisoquinolin-2(1H)-yl][(1S)-1- phenyl-3,4-dihydroisoquinolin-2(1H)-yl]methanone
CAS:2216750-52-4
Purity:≥98% Package:100mg
|
|
| | Solifenacin Impurity 7(Solifenacin EP Impurity D) Basic information |
| Product Name: | Solifenacin Impurity 7(Solifenacin EP Impurity D) | | Synonyms: | Solifenacin impurity 4/Solifenacin EP Impurity D/Solifenacin (R,S) Methanone Impurity/[(1R)-1-phenyl-3,4-dihydroisoquinolin-2(1H)-yl][(1S)-1-phenyl-3,4-dihydroisoquinolin-2(1H)-yl]methanone;[(1R)-1-Phenyl-3,4-dihydroisoquinolin-2(1H)-yl][(1S)-1-phenyl-3,4-dihydroisoquinolin-2(1H)-yl]methanone | | CAS: | 2216750-52-4 | | MF: | C31H28N2O | | MW: | 444.58 | | EINECS: | | | Product Categories: | | | Mol File: | 2216750-52-4.mol | 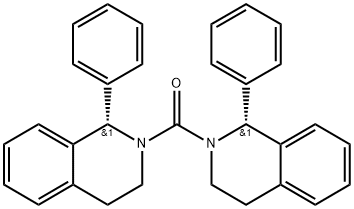 |
| | Solifenacin Impurity 7(Solifenacin EP Impurity D) Chemical Properties |
| Boiling point | 609.8±55.0 °C(Predicted) | | density | 1.200±0.06 g/cm3(Predicted) | | solubility | Chloroform (Sparingly), Ethyl Acetate (Slightly), Methanol (Slightly) | | form | Solid | | pka | -1.58±0.40(Predicted) | | color | White to Off-White |
| | Solifenacin Impurity 7(Solifenacin EP Impurity D) Usage And Synthesis |
| Uses | ((R)-1-phenyl-3,4-dihydroisoquinolin-2(1H)-yl)((S)-1-phenyl-3,4-dihydroisoquinolin-2(1H)-yl)methanone is a possible impurity of solifenacin (S676700) which is a muscarinic M3 receptor antagoinst. Solifenacin is used in treatment of urinary incontinence. |
| | Solifenacin Impurity 7(Solifenacin EP Impurity D) Preparation Products And Raw materials |
|