|
ChemicalBook Optimization Suppliers |
| 名前: |
BEST-REAGENT |
| 電話番号: |
400-1166-196 18981987031 |
| 電子メール: |
cdhxsj@163.com |
|
| | N-(ブトキシメチル)-2-クロロ-N-(2,6-ジエチルフェニル)アセトアミド 製品概要 |
| 化学名: | N-(ブトキシメチル)-2-クロロ-N-(2,6-ジエチルフェニル)アセトアミド | | 英語化学名: | Machette | | 别名: | N-(Butoxymethyl)-α-chloro-N(2,6-diethylphenyl)acetamide;N-(BUTOXYMETHYL)-2-CHLORO-2',6'-DIETHYLACETANILIDE;n-(butoxymethyl)-2-chloro-n-(2,6-diethylphenyl)acetamide;RASAYANCHLOR;TRAPP;n-(butoxymethyl)-2-chloro-n-(2,6-diethylphenyl)-acetamid;N-Butoxymethyl-.alpha.-chloro-2’,6’-diethylacetanilide;MACHETE | | CAS番号: | 23184-66-9 | | 分子式: | C17H26ClNO2 | | 分子量: | 311.85 | | EINECS: | 245-477-8 | | カテゴリ情報: | A-BAlphabetic;Alpha sort;B;BI - BZ;Pesticides&Metabolites;HERBICIDE;Agro-Chemicals | | Mol File: | 23184-66-9.mol | 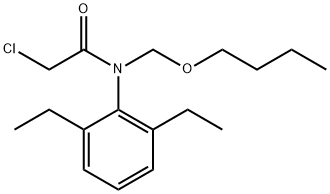 |
| | N-(ブトキシメチル)-2-クロロ-N-(2,6-ジエチルフェニル)アセトアミド 物理性質 |
| | N-(ブトキシメチル)-2-クロロ-N-(2,6-ジエチルフェニル)アセトアミド Usage And Synthesis |
| 溶解性 | 水:20mg/l(20℃)。ジエチルエーテル、アセトン、ベンゼン、エタノール、酢酸エチル、ヘキサンに可溶。エタノール及びアセトンに溶け、水にほとんど溶けない。 | | 用途 | ブタクロール試験用標準品。 | | 農薬用途 | 除草剤 | | 使用上の注意 | アルゴン封入 | | 化学的特性 | Yellow Oil | | 使用 | Butachlor is a pre-emergent chloroacetanalide herbicide. Butachlor is commonly used for weed control in rice as well as cotton, maize, wheat and other crops. | | 使用 | Pre-emergence selective anilide herbicide used
to control grass and broad-leaved weeds in seeded
and transplanted rice paddies. | | 定義 | ChEBI: Butachlor is an aromatic amide that is 2-choro-N-(2,6-diethylphenyl)acetamide in which the amide nitrogen has been replaced by a butoxymethyl group. It has a role as a herbicide, an environmental contaminant and a xenobiotic. It is an aromatic amide, an organochlorine compound and a tertiary carboxamide. It is functionally related to a N-phenylacetamide. | | 危険性 | Moderately toxic. | | 农业用途 | Herbicide: Used for pre-emergence control of annual grasses,
sedges and broadleaf weeds in rice crops. Used primarily
in Asia, South America, Europe and Africa. Not registered
for use in the U.S. Not approved for use in EU countries. | | 製品名 | BUTANEX®; BUTANOX®; CP 53619®;
HILTACHLOR®; LAMBAST®; MACHETE®;
MACHETTE®; PILLARSET®; RASAYANCHLOR®;
WEEDOUT®; VENDAVAL® | | 代謝経路 | In vitro incubation of butachlor with rat liver fractions
forms a considerable amount of glutathione conjugate,
while the conjugating activity is not efficient for the
kidney S9 fraction. Further biotransformation of the
glutathione conjugate to mercapturate is not observed
in the liver S9 fraction. Butachlor is initially conjugated
with glutathione in the liver and is apparently
transported to the kidneys where it is transformed to
mercapturic acid. |
|