|
|
| | Feruloylputrescine Basic information |
| Product Name: | Feruloylputrescine | | Synonyms: | Feruloylputrescine;N-(4-Aminobutyl)-3-(4-hydroxy-3-methoxyphenyl)propenamide;N-(4-Aminobutyl)-3-(4-hydroxy-3-methoxyphenyl)-2-propenamide;NSC 602818;N-(4-Aminobutyl)-3-(4-hydroxy-3-methoxyphenyl)acrylamide;2-Propenamide, N-(4-aminobutyl)-3-(4-hydroxy-3-methoxyphenyl)-;(E)-N-(4-aminobutyl)-3-(4-hydroxy-3-methoxyphenyl)prop-2-enamide;N-Feruloylputrescine | | CAS: | 501-13-3 | | MF: | C14H20N2O3 | | MW: | 264.32 | | EINECS: | | | Product Categories: | | | Mol File: | 501-13-3.mol | 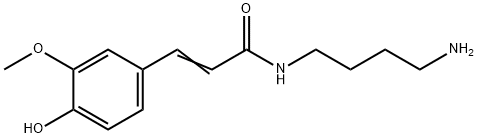 |
| | Feruloylputrescine Chemical Properties |
| Boiling point | 512℃ | | density | 1.156 | | Fp | 263℃ | | solubility | Water | | form | Solid | | pka | 9.78±0.35(Predicted) | | color | Yellowish | | LogP | 0.620 (est) |
| | Feruloylputrescine Usage And Synthesis |
| Description | Salsola subaphylla contains this alkaloid which is extremely hygroscopic and for
which no definite melting point has been obtained. It gives crystalline salts and
derivatives, e.g. the hydrochloride, melting over a very wide range of temperature;
the hydriodide which behaves similarly to the hydrochloride, picrate, m.p.
218.5°C. On hydrogenation over Pt02 it yields the dihydro derivative giving a
hydrochloride, m.p. 139-140oC and a picrate, m.p. 174.6-175°C. When boiled
with 30 per cent KOH it forms putrescine and ferulic acid and has the probable
structure given above. | | Uses | Feruloylputrescine is an effector of plant root systems, influencing development of nutrient gathering root systems. Inhibition of the formation of this compound causes the development of a dominant or ‘tap’ root. | | Definition | ChEBI: Subaphyllin is a hydroxycinnamic acid. | | References | Ryabinin, Il'na., Dokl. Akad. Nauk SSSR, 67, 513 (1949) |
| | Feruloylputrescine Preparation Products And Raw materials |
|