|
ChemicalBook Optimization Suppliers |
|
| | テトラブチルアンモニウムブロミド (IPC試薬) 製品概要 |
| 化学名: | テトラブチルアンモニウムブロミド (IPC試薬) | | 英語化学名: | Tetrabutylammonium bromide | | 别名: | n,n,n-tributyl-1-butanaminiubromide;Tetrabutylaminium·bromide;1-Butanaminium,N,N,N-tributyl-, bromide (1:1);Tetrabutylammonium b;TetrabutylaMMoniuM BroMide(Ipc-Tba-Br );TetrabutylaMMoniuM broMide, 99+% 100GR;TetrabutylaMMoniuM broMide, 99+% 25GR;TetrabutylaMMoniuM broMide, 99+% 500GR | | CAS番号: | 1643-19-2 | | 分子式: | C16H36BrN | | 分子量: | 322.37 | | EINECS: | 216-699-2 | | カテゴリ情報: | organo amine halide;Other Products;Ammonium SaltsEssential Chemicals;Reagent Plus;Routine Reagents;Ammonium SaltsAnalytical Reagents;Electrochemistry;Supporting Electrolytes for Electrochemistry;Ion Pair Reagents - Anionic Concentrate;AnionicHPLC;Chromatography/CE Reagents;Ion Pair;Ion Pair Reagents;Ion Pair Reagents - Anionic;Greener Alternatives: Catalysis;Phase Transfer Catalysts;quarternary ammonium salts;Ammonium Salts;Ammonium Bromides (Quaternary);Analytical Chemistry;HPLC Ion-Pair Reagents for Acidic Samples;Ion-Pair Reagents for HPLC;Quaternary Ammonium Compounds;Ammonium, Phosphonium, Sulfonium Salts (Ionic Liquids);Ionic Liquids;Synthetic Organic Chemistry;Pharmaceutical intermediates;1643-19-2;bc0001 | | Mol File: | 1643-19-2.mol | 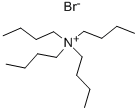 |
| | テトラブチルアンモニウムブロミド (IPC試薬) 物理性質 |
| 融点 | 102-106 °C(lit.) | | 沸点 | 102 °C | | 比重(密度) | 1.039 g/mL at 25 °C | | 蒸気圧 | 0Pa at 25℃ | | 屈折率 | n20/D 1.422 | | 闪点 | 100℃ | | 貯蔵温度 | Store below +30°C. | | 溶解性 | H2O: 0.1 g/mL, clear, colorless | | 酸解離定数(Pka) | 0[at 20 ℃] | | 外見 | Crystalline Powder | | 比重 | 1.007 | | 色 | White to slightly cream | | 臭い (Odor) | Amine like | | PH | 3.5 to 7.0 (50 g/L, 25 ℃) | | 水溶解度 | 600 g/L (20 ºC) | | Sensitive | Hygroscopic | | 極大吸収波長 (λmax) | λ: 240 nm Amax: 0.04
λ: 250 nm Amax: 0.03
λ: 260 nm Amax: 0.02
λ: 500 nm Amax: 0.02 | | BRN | 3570983 | | 安定性: | Stable. Incompatible with strong oxidizing agents. Protect from moisture. | | InChIKey | JRMUNVKIHCOMHV-UHFFFAOYSA-M | | LogP | 0.839 at 25℃ | | CAS データベース | 1643-19-2(CAS DataBase Reference) | | NISTの化学物質情報 | Tetra-N-butylammonium bromide(1643-19-2) | | EPAの化学物質情報 | Tetrabutylammonium bromide (1643-19-2) |
| | テトラブチルアンモニウムブロミド (IPC試薬) Usage And Synthesis |
| 外観 | 白色~ほとんど白色, 結晶~結晶性粉末 | | 溶解性 | 水, アルコールに可溶。水及びエタノールによく溶け、アセトンに溶けやすい。 | | 用途 | イオンペアクロマトグラフでの酸性物質のカウンターイオン剤。有機合成(和光試薬時報Vol.64 No.2,p.26(1996))。 | | 用途 | 産業用インクジェットプリンター用インク
| | 使用上の注意 | 吸湿性あり | | 化学的特性 | white crystals or powder | | 使用 | Tetrabutylammonium Bromide is used in the synthesis of polymer solar cells. Also used in the synthesis of single component green-light emitting electrochemical cells. | | 定義 | ChEBI: Tetrabutylammonium bromide is a tetrabutylammonium salt with bromide as the anionic counterpart. It is an organic bromide salt and a tetrabutylammonium salt. | | 利点 | Tetrabutylammonium bromide is an environmentally benign, non-volatile, non-flammable, non-corrosive, low-cost, commercially available ammonium salt with high thermal and chemical stability. In TBAB, tetrabutylammonium salt can dissolve in both aqueous as well as in organic solvents, which helps to transport the water-soluble anionic reactants into the organic phase. Moreover, molten TBAB was also employed as an efficient ionic liquid to carry out organic transformations under solvent-free conditions[1]. | | 一般的な説明 | Tetrabutylammonium bromide, a quaternary ammonium compound widely used as a phase transfer catalyst. TBAB decreases the retention time and removes peak tailing by acting as an ion pair reagent during the chromatographic analysis of quaternary ammonium compounds. In the molten state, TBAB behaves like an ionic liquid, which is a promising green alternative to organic solvents in polymer synthesis. | | 燃焼性と爆発性 | Not classified | | 純化方法 | Crystallise the salt from *benzene (5mL/g) at 80o by adding hot n-hexane (three volumes) and allowing to cool. Dry it over P2O5 or Mg(ClO4)2, under vacuum. The salt is very hygroscopic. It can also be crystallised from ethyl acetate or dry acetone by adding diethyl ether and dried in vacuo at 60o for 2 days. It has been crystallised from acetone by addition of diethyl ether. It is so hygroscopic that all manipulations should be carried out in a dry-box. It has been purified by precipitation from a saturated solution in dry CCl4 on addition of cyclohexane or by recrystallisation from ethyl acetate, then heating in vacuum to 75o in the presence of P2O5. [Symons et al. J Chem Soc, Faraday Trans 1 76 2251 1908.] It also recrystallises from CH2Cl2/diethyl ether and is dried in a vacuum desiccator over P2O5. [Blau & Espenson J Am Chem Soc 108 1962 1986, Beilstein 4 IV 657.] | | 参考文献 | [1] Banik B, et al. Tetrabutylammonium Bromide (TBAB) Catalyzed Synthesis of Bioactive Heterocycles. Molecules, 2020; 25: 5918. |
|