|
ChemicalBook Optimization Suppliers |
|
| 融点 | <25℃ | | 比旋光度 | D -8 to -9° (chloroform) | | 沸点 | 129-130 °C/14 mmHg (lit.) | | 比重(密度) | 0.901 g/mL at 25 °C (lit.) | | 蒸気圧 | 6Pa at 20℃ | | FEMA | 2252 | BETA-CARYOPHYLLENE | | 屈折率 | n20/D 1.5(lit.) | | 闪点 | 205 °F | | 貯蔵温度 | -20°C | | 溶解性 | Chloroform (Sparingly), DMSO (Slightly), Methanol (Slightly) | | 色 | Colourless | | 比重 | 0.90 | | 臭い (Odor) | at 100.00 %. sweet woody spice clove dry | | においのタイプ | spicy | | 光学活性 (optical activity) | [α]23/D 7.5°, neat | | 水溶解度 | 88μg/L at 20℃ | | Merck | 14,1875 | | JECFA Number | 1324 | | BRN | 2044564 | | 安定性: | Light Sensitive | | LogP | 6.23 at 25℃ | | CAS データベース | 87-44-5(CAS DataBase Reference) | | EPAの化学物質情報 | Bicyclo[7.2.0]undec-4-ene, 4,11,11-trimethyl-8-methylene-, (1R,4E,9S)- (87-44-5) |
| Rフレーズ | 36/37/38 | | Sフレーズ | 26-36-24/25 | | RIDADR | UN1230 - class 3 - PG 2 - Methanol, solution | | WGK Germany | 1 | | RTECS 番号 | DT8400000 | | HSコード | 29021990 |
| | β-カリオフィレン Usage And Synthesis |
| 外観 | 無色~うすい黄色、液体 | | 定義 | 本品は、次の化学式で表されるセスキテルペン炭化水素である。 | | 解説 | (1R-4E-9S)-4,11,11-trimethyl-8-methylenebicyclo[7.2.0]undec-4-ene.C15H24(204.34).β-カリオフィレンともいう.二環性セスキテルペン.フトモモ科Eugenis caryophyllaのクローブ油のなかにα-カリオフィレンとともに含まれているが,アカマツPinns densifloraやヤマジソMosla japonicaなど,植物界に広く存在している.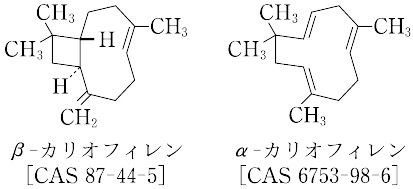 "沸点129~130 ℃(1.9 kPa).[α]D"-9.9°(ベンゼン).2分子の塩化水素を付加して融点69~70 ℃ の結晶になる.芳香性材料として使われる.α-カリオフィレンはα-フムレンと同じ. "沸点129~130 ℃(1.9 kPa).[α]D"-9.9°(ベンゼン).2分子の塩化水素を付加して融点69~70 ℃ の結晶になる.芳香性材料として使われる.α-カリオフィレンはα-フムレンと同じ.
森北出版「化学辞典(第2版) | | 用途 | 有機合成原料。 | | 化粧品の成分用途 | 皮膚コンディショニング剤、香料 | | 説明 | β-Caryophyllene (Item No. 21572) is a sesquiterpene that has been found in plants, including C. sativa, C. indica, and hemp, and has diverse biological activities, including lipid metabolic, antioxidant, anti-neuroinflammatory, anti-proliferative, and antinociceptive properties. It is an agonist of the cannabinoid (CB) receptor CB2 (Ki = 155 nM) that inhibits cAMP production induced by forskolin in CHO-K1 cells expressing CB2 receptors (EC50 = 38 nM). β-Caryophyllene is also an agonist of peroxisome proliferator-activated receptor α (PPARα; EC50 = 9.58 μM in a reporter assay). β-Caryophyllene (1 and 2.5 μM) reduces the production of reactive oxygen species (ROS) in and protects against cytotoxicity of SH-SY5Y cells induced by 1-methyl-4-pheylpyridinium (MPP+). It also decreases the β-amyloid burden in the hippocampus and cerebral cortex and improves memory in an APP/PS1 transgenic mouse model of Alzheimer’s disease, decreasing the latency to find the platform in the Morris water maze during training and increasing the time spent in the target quadrant during testing when administered at a dose of 48 mg/kg per day. β-Caryophyllene (50 mg/kg) increases the number of entries into and the time spent in the open arms of the elevated plus maze and the time spent immobile in the forced swim test, indicating anxiolytic-like and antidepressant-like activity, effects that can be blocked by the CB2 receptor antagonist AM630 . | | 化学的特性 | Clear colorless liquid | | 化学的特性 | β-Caryophyllene has a woody-spicy, dry, clove-like aroma. | | 天然物の起源 | Three isomers (α-, β-, and γ-caryophyllene) are found in nature. The β-isomer is the most frequently encountered
and most abundant. This sesquiterpene hydrocarbon occurs naturally in approximately 60 essential oils, mainly in that of
cloves, from which it was originally isolated. The chemical structure has been thoroughly studied; other studies have been conducted
on the isolation and the dipolar moment, as well as on its oxide. Reported found in lime peel oil, lemon, grapefruit, guava fruit, raspberry,
black currant, carrot, celery seed, cinnamon bark, anise, nutmeg, cumin seed, ginger, pepper, peppermint oil, mace, laurel and
caraway herb. | | 使用 | β-Caryophyllene is notable for having a cyclobutane ring, a rarity in nature. β-Caryophyllene is one of the chemical compounds that contributes to the spiciness of black pepper. β-Caryophyllene was shown to selectively bind to the cannabinoid receptor type-2 (CB2) and to exert significant cannabimimetic antiinflammatory effects in mice. | | 製造方法 | Isolated from oil of clove stems and separated from eugenol by treating the oil with 7% sodium carbonate solution,
extracting with ether, repeating the carbonate treatment on the concentrated extracts, and finally steam distilling. | | 定義 | ChEBI: A beta-caryophyllene in which the stereocentre adjacent to the exocyclic double bond has S configuration while the remaining stereocentre has R configuration. It is the most commonly occurring form of <greek beta-caryophyllene, occurring in many essential oils, particularly oil of cloves. | | Aroma threshold values | Detection at 64 to 90 ppb | | Taste threshold values | Taste characteristics at 50 ppm: spicy, pepper-like, woody, camphoraceous with a citrus background. | | Synthesis Reference(s) | Journal of the American Chemical Society, 86, p. 485, 1964 DOI: 10.1021/ja01057a040 | | 一般的な説明 | Pale yellow oily liquid with an odor midway between odor of cloves and turpentine. | | 空気と水の反応 | Insoluble in water. | | 反応プロフィール | The unsaturated aliphatic hydrocarbons, such as BETA-CARYOPHYLLENE, are generally much more reactive than the alkanes. Strong oxidizers may react vigorously with them. Reducing agents can react exothermically to release gaseous hydrogen. In the presence of various catalysts (such as acids) or initiators, compounds in this class can undergo very exothermic addition polymerization reactions. | | 火災危険 | BETA-CARYOPHYLLENE is combustible. | | 安全性プロファイル | A skin irritant.
Combustible liquid. When heated to
decomposition it emits acrid smoke and
irritating fumes. | | 発がん性 | Caryophyllene showed significant
activity as an inducer of the detoxifying enzyme glutathione
S-transferase in the mouse liver and small intestine.
The ability of natural anticarcinogens to induce detoxifying
enzymes has been found to correlate with their activity in the
inhibition of chemical carcinogenesis (253a). |
|