2,6-DIMETHYLPYRIDINE-4-CARBOXALDEHYDE manufacturers
|
| | 2,6-DIMETHYLPYRIDINE-4-CARBOXALDEHYDE Basic information |
| Product Name: | 2,6-DIMETHYLPYRIDINE-4-CARBOXALDEHYDE | | Synonyms: | 2,6-DIMETHYLPYRIDINE-4-CARBOXALDEHYDE;2,6-Dimethylisonicotinaldehyde;2,6-diMethyl-4-Pyridinecarboxaldehyde;6-diMethylpyridine-4-carbaldehyde;2,6-DiMethylpyridine-4-carbaldehyde;4-Pyridinecarboxaldehyde, 2,6-dimethyl-;2,6-Dimethylpyridin-4-carbaldehyde | | CAS: | 18206-06-9 | | MF: | C8H9NO | | MW: | 135.16 | | EINECS: | | | Product Categories: | Pyridines | | Mol File: | 18206-06-9.mol | 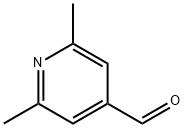 |
| | 2,6-DIMETHYLPYRIDINE-4-CARBOXALDEHYDE Chemical Properties |
| Melting point | 8℃ | | Boiling point | 218℃ | | density | 1.065 | | refractive index | 1.5282 | | Fp | 91℃ | | storage temp. | 2-8°C, stored under nitrogen | | pka | 4.69±0.10(Predicted) | | Appearance | Colorless to light yellow Liquid |
| | 2,6-DIMETHYLPYRIDINE-4-CARBOXALDEHYDE Usage And Synthesis |
| Synthesis | The general procedure for the synthesis of 2,6-dimethylpyridine-4-carbaldehyde from 2,6-dimethyl-4-hydroxymethylpyridine was as follows: 2,6-dimethyl-4-hydroxymethylpyridine (1.00 g, 7.30 mmol) was dissolved in acetonitrile (20 mL), and 1,2-benzoyl-3(1H)-keto-1-hydroxyl-1-oxo (IBX) (2.00 g, 7.30 mmol). The reaction mixture was heated to reflux for 6 hours. After completion of the reaction, the solid was removed by filtration and washed with acetonitrile. The filtrate was concentrated under reduced pressure to give 2,6-dimethylpyridine-4-carbaldehyde as a brown liquid. Yield: 0.81 g (82%). | | References | [1] Patent: US9238640, 2016, B2. Location in patent: Page/Page column 134; 135
[2] Patent: US2013/281399, 2013, A1. Location in patent: Paragraph 0732; .0735; 0740; 0760; 0770; 0752
[3] Chemical Communications, 2015, vol. 51, # 61, p. 12258 - 12261 |
| | 2,6-DIMETHYLPYRIDINE-4-CARBOXALDEHYDE Preparation Products And Raw materials |
|