|
|
| | Albuterol Aldehyde Basic information |
| Product Name: | Albuterol Aldehyde | | Synonyms: | Albuterol Aldehyde;5-[(1RS)-2-[(1,1-diMethylethyl)aMino]-1-hydroxyethyl]-2-hydroxybenzaldehyde;5-(2-tert-Butylamino-1-hydroxyethyl)-2-hydroxybenzaldehyde;Levalbuterol Related Compound D (30 mg) (5-[2-{(1,1-dimethylethyl)amino}-1-hydroxyethyl]-2-hydroxy-benzaldehyde sulfate);Levalbuterol Related CoMpound D;Salbutamol Impurity D(EP)(hydrochloride);Levalbuterol Related Compound D (Salbutamol EP Impurity D/ Salbutamol Aldehyde Impurity);Salbutamol EP Impurity D (Salbutamol Aldehyde Impurity/ Levalbuterol Related Compound D) | | CAS: | 156339-88-7 | | MF: | C13H19NO3 | | MW: | 237.29 | | EINECS: | 604-604-1 | | Product Categories: | Amines;Aromatics;Impurities;Intermediates & Fine Chemicals;Pharmaceuticals;DCP | | Mol File: | 156339-88-7.mol | 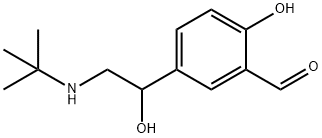 |
| | Albuterol Aldehyde Chemical Properties |
| Melting point | 200-210°C | | Boiling point | 392.4±42.0 °C(Predicted) | | density | 1.154±0.06 g/cm3(Predicted) | | storage temp. | 2-8°C | | solubility | DMSO (Slightly, Heated), Water (Slightly), Methanol | | form | solid | | pka | 8.02±0.31(Predicted) | | Major Application | pharmaceutical | | InChI | InChI=1S/C13H19NO3/c1-13(2,3)14-7-12(17)9-4-5-11(16)10(6-9)8-15/h4-6,8,12,14,16-17H,7H2,1-3H3 | | InChIKey | RNGYKBNYRUYCIV-UHFFFAOYSA-N | | SMILES | C(=O)C1=CC(C(O)CNC(C)(C)C)=CC=C1O |
| WGK Germany | WGK 3 | | HS Code | 2922504500 | | Storage Class | 11 - Combustible Solids | | Hazard Classifications | Eye Irrit. 2
Skin Irrit. 2
STOT SE 3 |
| | Albuterol Aldehyde Usage And Synthesis |
| Chemical Properties | Pale Yellow Solid | | Uses | A related impurity of Albuterol (A514500). | | Synthesis | 1.0g of salbutamol, dissolved in 20ml of N,N-dimethylformamide, then added 10% molar equivalent of tetramethylpiperidine oxide, 10% molar equivalent of tetrabutylammonium chloride, and 20ml of 0.5 mol/L aqueous NaHCO3 solution, after stirring at room temperature for 30min, added 1.1 molar equivalent of N-chlorosuccinimide, after reacting at room temperature for 2~5h After the reaction, most of the solvent was removed, extracted with ethyl acetate, washed, dried, and purified by column chromatography after removing the solvent, a white solid salbutamol monoxide was obtained as salbutamol impurity D.
|
| | Albuterol Aldehyde Preparation Products And Raw materials |
|