|
ChemicalBook Optimization Suppliers |
|
| 化学名: | ブメトリゾール | | 英語化学名: | UV Absorber 326 | | 别名: | 2-T-BUTYL-6-(5-CHLORO-2 H-BENZOTRIAZOL-2-YL)-4-METHYLPHENOL;LABOTEST-BB LT00138024;2-(5-chloro-2h-benzotriazol-2-yl)-6-(1,1-dimethylethyl)-4-methyl-pheno;Phenol, 2-(5-chloro-2H-benzotriazol-2-yl)-6-(1,1-dimethylethyl)-4-methyl-;ACETOSTAB 326;2-(5-CHLORO-2-BENZOTRIAZOLYL)-6-TERT-BUTYL-PARA-CRESOL;2-(5-Chlor-2H-benzotriazol-2-yl)-6-(1,1-dimethylethyl)-4-methyl-phenol;Tinuvin 326 Ciba | | CAS番号: | 3896-11-5 | | 分子式: | C17H18ClN3O | | 分子量: | 315.8 | | EINECS: | 223-445-4 | | カテゴリ情報: | Polymer Additives;Polymer Science;Polymer Additives;Polymer Science;Stabilizers;Industrial/Fine Chemicals;3896-11-5 | | Mol File: | 3896-11-5.mol | 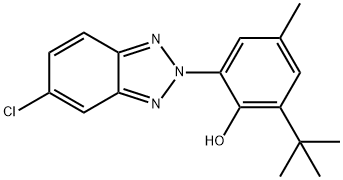 |
| 融点 | 144-147 °C(lit.) | | 沸点 | 460.4±55.0 °C(Predicted) | | 比重(密度) | 1.26±0.1 g/cm3(Predicted) | | 蒸気圧 | 0Pa at 20℃ | | 貯蔵温度 | Sealed in dry,Room Temperature | | 溶解性 | Chloroform (Slightly), Ethyl Acetate (Slightly), Methanol (Slightly) | | 酸解離定数(Pka) | 9.31±0.48(Predicted) | | 外見 | Solid | | 色 | Pale Yellow to Light Yellow | | 水溶解度 | 4μg/L at 20℃ | | InChI | InChI=1S/C17H18ClN3O/c1-10-7-12(17(2,3)4)16(22)15(8-10)21-19-13-6-5-11(18)9-14(13)20-21/h5-9,22H,1-4H3 | | InChIKey | OCWYEMOEOGEQAN-UHFFFAOYSA-N | | SMILES | C1(O)=C(C(C)(C)C)C=C(C)C=C1N1N=C2C=C(Cl)C=CC2=N1 | | LogP | 6.580 (est) | | CAS データベース | 3896-11-5(CAS DataBase Reference) | | EPAの化学物質情報 | 2-tert-Butyl-6-(5-chloro-2H-benzotriazol-2-yl)-p-cresol (3896-11-5) |
| | ブメトリゾール Usage And Synthesis |
| 外観 | 白色~うすい黄色, 結晶性粉末~粉末 | | 定義 | 本品は、次の化学式で表される複素環式化合物である。 | | 溶解性 | メチルエチルケトン、トルエン、酢酸エチル、スチレンモノマー、ジオクチルフタレートに可溶。 | | 用途 | 紫外線吸収剤。 | | 化粧品の成分用途 | 褪色防止剤 | | 効能 | 紫外線保護剤 | | 化学的特性 | Pale Yellow Solid | | Originator | Bumetrizole,Onbio Inc. | | 使用 | UV-326 can potentially be used as a UV light filter in cosmetics and sunscreens. It may also be used as a corrosion inhibitor for the protection of building materials and automobiles. | | 使用 | Bumetrizole is an intermediate reactant in the synthesis of UV light absorbers for polyester fibers. | | Manufacturing Process | To a 2000 ml 3-necked, round-bottomed flask equipped with an agitator,
reflux condenser, nitrogen inlet and thermometer were charged 140.5 g of 2'-
hydroxy-3'-t-butyl-5'-methyl-5-chloro-2-nitroazobenzene, 119 g of isopropanol
and 80 g of Amsco mineral spirits. A stream of nitrogen was introduced over
the surface of the contents of the flask and the nitrogen atmosphere was then
maintained throughout the remainder of the reduction process. 13.7 g of 50%
aqueous sodium hydroxide solution and 222 g of water were added and the
temperature of the contents of the flask were adjusted to 55°C. The ratio of
the moles of alkali to moles of o-nitroazobenzene intermediate used was is
0.848/1. 104 g of zinc dust was added in 5 portions over a 2 hours period
with the temperature of the flask being held at 55-60°C with some slight
external cooling. The total concentration of iron impurities from all reactants
less than 150 ppm based on zinc used. After all the zinc was added, the
temperature was raised to 60°C and held at this temperature until a spot test
indicated no more o-nitroazobenzene intermediate was present. The
temperature was then raised to 65°C and held there for 4 to 5 hours or until
TLC analysis indicated that no more of the N-oxy-intermediate was present.
62.6 g of anhydrous sodium sulfate and 35.6 g of water were then added, the
batch was heated to 70°C and stirred for 15 min. The material was then
allowed to stand and separate into three liquid phases plus unreacted zinc
dust. The top two layers containing the desired product were transferred to
another flask. The remaining aqueous zinc slurry was washed at 65-70°C with
three successive 16 g portions of Amsco mineral spirits: isopropanol 50:50.
The combined product layers and wash liquids were then washed once at 70°C
with an aqueous hydrochloric acid solution made from 130 g of water and 40
g of 32% hydrochloric acid to remove cleavage amine by-products. A second
and third wash followed at 70°C with aqueous hydrochloric acid solutions
made each from 65 g of water and 20 g of 32% hydrochloric acid. The last
wash was essentially colorless. 14 g of 32% hydrochloric acid and 220 g of
isopropanol were added to the solution of the product. The batch was allowed
to crystallize slowly. The solid product form was filtered and washed with
isopropanol at 0°C to give 110 g of 5-chloro-2-(2-hydroxy-3-t-butyl-5-
methylphenyl)-2H-benzotriazole with a melting point of 140-141°C. | | Therapeutic Function | Sunscreen agent | | 一般的な説明 | 2-tert-Butyl-6-(5-chloro-2H-benzotriazol-2-yl)-4-methylphenol (UV-326) is a benzotriazole based UV absorber that can be used in the photostabilization of industrial materials. It can absorb the harmful UV radiation for the wavelength of about 290 nm. | | 燃焼性と爆発性 | Not classified |
|