|
ChemicalBook Optimization Suppliers |
| 名前: |
Alfa Aesar |
| 電話番号: |
400-6106006 |
| 電子メール: |
saleschina@alfa-asia.com |
| 名前: |
Energy Chemical |
| 電話番号: |
021-021-58432009 400-005-6266 |
| 電子メール: |
sales8178@energy-chemical.com |
|
| | 重クロム酸ピリジニウム Usage And Synthesis |
| 外観 | 黄赤色~褐色粉末~結晶 | | 解説 | 重クロム酸ピリジニウム融点144~146 ℃.水,ジメチルホルムアミド,ジメチルスルホキシドに可溶.緩和な酸化剤で,ジメチルホルムアミド中では,アルデヒドや第一級アルコールをカルボン酸にするが,ジクロロメタン中では,第一級アルコールはアルデヒドになる. 森北出版「化学辞典(第2版)
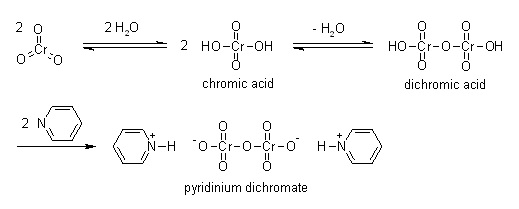
| | 用途 | コーンフォース試薬は第一級および第二級アルコールをアルデヒドおよびケトンにそれぞれ酸化する強力な酸化剤である。化学構造および機能性は、クロロクロム酸ピリジニウムやコリンズ試薬などその他酸化六価クロム化合物と密接に関連している。 | | 製造 | 重クロム酸ピリジニウム略称PDC.三酸化クロムを少量の水に溶かし,ピリジンを加えてつくった塩で,オレンジ色の結晶. | | 化学的特性 | Orange solid | | 使用 | Pyridinium dichromate (PDC) is an orange colored solid used as an oxidizing agent.
Oxidizing agent for conversion of primary alcohols to aldehydes and ketones, acetals to esters, and didehydroketones to enones in the presence of tert-butyl hydroperoxide.
Pyridinium Dichromate may be used as an alternative to PCC in nucleoside and carbohydrate oxidation, particularly for fragile molecules. PDC can also be used in conjunction with tertbutylhydroperoxide for a variety of oxidative transformations. | | 使用 | Pyridinium dichromate acts as a strong oxidizing agent used in the conversion of primary alcohols and secondary alcohols to aldehydes and ketones respectively. It plays an important role in the oxidation of unsaturated tertiary alcohols, silyl ethers, the carbon-boron bond, and oximes. Further, it is used in the conversion of acetals to esters and didehydroketones to enones in the presence of tert-butyl hydroperoxide. | | 定義 | ChEBI: Pyridinium dichromate is a pyridinium salt that is the dipyridinium salt of dichromic acid. It is a strong oxidizing agent which can convert primary and secondary alcohols to aldehydes and ketones respectively. It has a role as an oxidising agent. It contains a dichromate(2-). | | 製造方法 | Pyridinium dichromate can be obtained by gradual addition of a solution of chromic anhydride (Cr2O3) in water to pyridine in ice cold conditions.
 | | 燃焼性と爆発性 | Flammable | | 純化方法 | Dissolve it in the minimum volume of H2O and add 5 volumes of cold Me2CO and cool to -20o. After 3hours the orange crystals are collected, washed with a little cold Me2CO and dried in a vacuum. It is soluble in dimethylformamide (0.9g/mL at 25o), and in H2O, and has a characteristic IR with �max at 930, 875, 765, 730 and 730 cm-1. [Corey & Schmidt Tetrahedron Lett 399 1979, Coats & Corrigan Chem Ind (London) 1594 1969.] (Possible CARCINOGEN.) § Available commercially on a polymer support. |
|