| | 2-NITROBENZYL CHLORIDE Basic information |
| Product Name: | 2-NITROBENZYL CHLORIDE | | Synonyms: | 1-Nitro-2-(chloromethyl)benzene;2-(Chloromethyl)nitrobenzene, alpha-Chloro-2-nitrotoluene;2-Nitrobenzyl chloride 99%;2-NITROBENZYL CHLORIDE;A-CHLORO-O-NITROTOLUENE;2-NitrobenzylChloride98%;à-chloro-2-nitrotoluene;α-chloro-2-nitrotoluene | | CAS: | 612-23-7 | | MF: | C7H6ClNO2 | | MW: | 171.58 | | EINECS: | 210-300-5 | | Product Categories: | | | Mol File: | 612-23-7.mol | 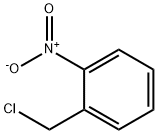 |
| | 2-NITROBENZYL CHLORIDE Chemical Properties |
| Melting point | 46-48 °C (lit.) | | Boiling point | 127-133 °C/10 mmHg (lit.) | | density | 1,556 g/cm3 | | refractive index | 1.5557 (estimate) | | Fp | >230 °F | | storage temp. | Store below +30°C. | | solubility | ethanol: soluble98% | | form | Crystals, Crystalline Powder or Chunks | | color | Yellow to tan | | Water Solubility | Soluble in acetone, alcohol benzene, diethyl ether, and ethanol (98%). Insoluble in water. | | BRN | 388396 | | Stability: | Stable. Incompatible with bases, moisture, acohols, oxidizing agents, amines. Corrodes steel. | | CAS DataBase Reference | 612-23-7(CAS DataBase Reference) | | EPA Substance Registry System | o-Nitrobenzyl chloride (612-23-7) |
| Hazard Codes | C | | Risk Statements | 34-36 | | Safety Statements | 26-36/37/39-45-27 | | RIDADR | UN 3261 8/PG 2 | | WGK Germany | 3 | | RTECS | XS9092000 | | F | 19-21 | | HazardClass | 8 | | PackingGroup | II | | HS Code | 29049090 |
| | 2-NITROBENZYL CHLORIDE Usage And Synthesis |
| Chemical Properties | liquid | | Uses | 2-Nitrobenzyl chloride was used in the synthesis of S-(2-nitrobenzyl)cysteine which was inserted into human erythropoietin. 2-Nitrobenzyl chloride was used to prepare 4,4′-dinitrostillbene. | | Uses | 2-Nitrobenzyl chloride has been used to introduce the photo labile 2-nitrobenzyl protecting group for the phenolic OH group of tyrosine. It is used as a pharmaceutical intermediate. | | General Description | Pale yellow crystals. Insoluble in water. Is thermally unstable. | | Air & Water Reactions | Insoluble in water. | | Reactivity Profile | A halogenated aromatic nitro compound. Aromatic nitro compounds range from slight to strong oxidizing agents. If mixed with reducing agents, including hydrides, sulfides and nitrides, they may begin a vigorous reaction that culminates in a detonation. The aromatic nitro compounds may explode in the presence of a base such as sodium hydroxide or potassium hydroxide even in the presence of water or organic solvents. The explosive tendencies of aromatic nitro compounds are increased by the presence of multiple nitro groups. | | Health Hazard | ACUTE/CHRONIC HAZARDS: Toxic. Lachrymator. Causes irritation on contact. Hazardous decomposition products. Mutagen. | | Fire Hazard | 2-NITROBENZYL CHLORIDE is combustible. | | Biochem/physiol Actions | 2-Nitrobenzyl chloride reacts with N-methyldiethanolamine and further reaction of the resulting quaternary diol with thionyl chloride at room temperature yields nitrobenzyl mustard quaternary salts. |
| | 2-NITROBENZYL CHLORIDE Preparation Products And Raw materials |
|