|
ChemicalBook Optimization Suppliers |
|
| 化学名: | シクロスポリン | | 英語化学名: | Cyclosporin A | | 别名: | ol27-400;s7481f1;sandimmun;Cyclospori;1,4,7,10,13,16,19,22,25,28,31-Undecaazacyclotritriacontane, cyclic peptide deriv.;30-Ethyl-33-(1-hydroxy-2-methyl-hex-4-enyl)-1,4,7,10,12,15,19,25,28-nonamethyl-6,9,18,24-tetrakis(2-methylpropyl)-3,21-dipropan-2-yl-1,4,7,10,13,16,19,22,25,28,31-undecazacyclotritriacontane-2,5,8,11,14,17,20,23,26,29,32-undecone;Cipol N;Neoplanta | | CAS番号: | 59865-13-3 | | 分子式: | C62H111N11O12 | | 分子量: | 1202.61 | | EINECS: | 611-907-1 | | カテゴリ情報: | Signalling;Protein Phosphatase;KAYQUINONE;antibiotic;Anti-cancer&immunity;Inhibitors;Intermediates & Fine Chemicals;Peptides;Pharmaceuticals;Active Pharmaceutical Ingredients;Organics;APIs;1 | | Mol File: | 59865-13-3.mol | 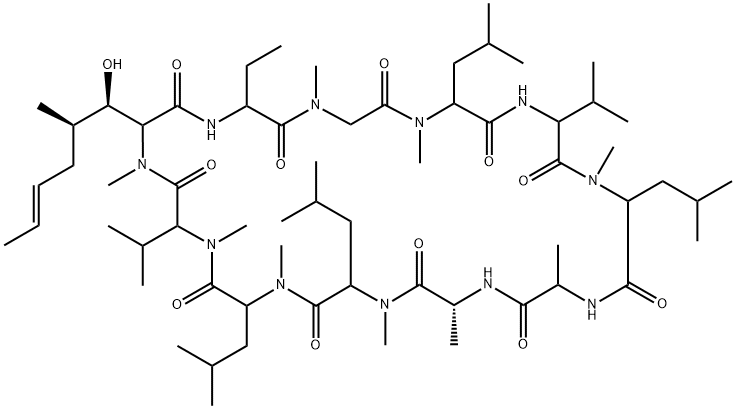 |
| 融点 | 148-151°C | | 沸点 | 838.63°C (rough estimate) | | 比旋光度 | D20 -244° (c = 0.6 in chloroform); D20 -189° (c = 0.5 in methanol) | | 比重(密度) | 0.9913 (rough estimate) | | 屈折率 | 1.6500 (estimate) | | 闪点 | 87℃ | | 貯蔵温度 | -20°C | | 溶解性 | ethanol: 30 mg/mL | | 外見 | solid | | 酸解離定数(Pka) | 13.32±0.70(Predicted) | | 色 | white | | 水溶解度 | Soluble in dimethyl sulfoxide and ethanol. Insoluble in water. | | Merck | 14,2752 | | BRN | 3647785 | | 安定性: | Stable for 3 years as supplied. Solutions in DMSO or ethanol may be stored at -20°C for up to 3 months. | | InChIKey | PMATZTZNYRCHOR-CGLBZJNRSA-N | | CAS データベース | 59865-13-3(CAS DataBase Reference) | | IARC | 1 (Vol. 50, 100A) 2012 | | EPAの化学物質情報 | Cyclosporin A (59865-13-3) |
| | シクロスポリン Usage And Synthesis |
| 外観 | 白色~わずかにうすい褐色、結晶性粉末~粉末、又は塊 | | 溶解性 | エタノール(95)に極めて溶けやすく、アセトンに溶けやすく、水にほとんど溶けない。 | | 解説 | かび由来の環状ペプチドで,免疫抑制作用がある.
森北出版「化学辞典(第2版) | | 用途 | シクロスポリン(Ciclosporin(INN?BAN(英語版))またはサイクロスポリン(Cyclosporine(USAN)、Cyclosporin(旧BAN))とは、真菌が産生する環状ポリペプチド抗生物質の一つである。D-アミノ酸を1つ含む11のアミノ酸からなる。
Tリンパ球によるインターロイキン-2,4,5,13やインターフェロン-γなどのサイトカイン転写を特異的かつ可逆的に抑制し、ひいてはサイトカイン産生と遊離を抑制する。これはカルシニューリンによる細胞内情報伝達阻害による。臓器移植による拒絶反応の抑制や自己免疫疾患の治療に使用される。 | | 用途 | 11 個のアミノ酸から成る環
状ポリペプチドです、免疫反応を促すヘル
パー T 細胞からのインターロイキン 2 の分泌
を阻害して免疫抑制作用を示します。 | | 効能 | 免疫抑制薬, カルシニューリン阻害薬 | | 商品名 | サンディミュン (ノバルティスファーマ); サンディミュン (ノバルティスファーマ); ネオーラル (ノバルティスファーマ); パピロック (参天製薬) | | 説明 | Cyclosporine A is a powerful immunosuppressive drug intended for preventing rejection
of kidney, heart, and lung transplants.
A new era in the development of immunopharmacology began with the discovery of
cyclosporines.
Cyclosporines are produced by mycelial mushrooms Tolypocladium inflatum,
Tricoderma polysporum, and Cylindrocarpon lucidum, which are found in the ground.
Cyclosporine A is the first drug to affect a specific line of protecting cells of the body.
Unlike usual cytotoxics, it suppresses T-cells and acts on all cell lines simultaneously.
Cyclosporine A significantly eases the ‘reception’ of transplants, and increases the possi�bility of treating autoimmune system diseases. | | 化学的特性 | White crystalline solid or powder. | | 化学的特性 | White or almost white powder | | Originator | Sandimmune,Sandoz,US,1983 | | 使用 | An immunosuppressant that has revolutionized organ transplantation through its use in the prevention of graft rejection.
A group of nonpolar cyclic oligopeptides with immunosupppressant activity. | | 使用 | prothrombogenic agent | | 使用 | Cyclosporin A is a hydrophobic cyclic peptide isolated from several fungal species including Cylindrocarpon, Fusarium, Trichoderma and Tolypocladium. Cyclosporin A inhibits T-cell activation and has been marketed since 1983 as an immunosuppressant in post-allogeneic organ transplant. Cyclosporin A acts by binding to the protein, cyclophilin (immunophilin), in T-lymphocytes causing inhibition of calcineurin (protein phosphatase 2B). Cyclosporin A reduces transcription of interleukin 2, and inhibits lymphokine production, interleukin release and NO synthesis induced by interleukin 1α, lipopolysaccharides and TNFα. | | 定義 | ChEBI: A cyclic nonribosomal peptide of eleven amino acids; an immunosuppressant drug widely used in post-allogeneic organ transplant to reduce the activity of the patient's immune system, and therefore the risk of organ rejection. Also causes reversible inhibiti
n of immunocompetent lymphocytes in the G0- and G1-phase of the cell cycle. | | 適応症 | | | Manufacturing Process | 10 liters of a nutrient solution (of which each liter contains 30 g of sucrose,
10 g of corn steep, 3 g of NaNO3, 1 g of K2HPO4, 0.5 g of MgSO4·7H2O, 0.5 g
of KCl and 0.01 g of FeSO4·7H2O) are inoculated with 100 cc of a conidia and mycelium suspension of the strain NRRL 5760, and incubation is effected in
700 cc penicillin flasks at 27°C for 11 days.
The mycelium, which has been separated from the culture liquid, is extracted
in a Turrax apparatus by crushing and stirring with 3.5 liters of 90%
methanol, and the crushed mycelium, which is separated from the solvent by
filtering with suction, is again treated twice in the same manner with 90%
methanol. The combined filtrates are concentrated by evaporation in a
vacuum at a bath temperature of 40°C to such an extent that the vapor
mainly consists of water alone. The resulting mixture is extracted six times
with the same volume of ethylene chloride by shaking, whereupon the
combined ethylene chloride solutions are purified by extraction with water and
are concentrated by evaporation in a vacuum at a bath temperature of 40°C.
The resulting residue is chromatographed on 250 g of silica gel (silica gel 60
Merck, grain size 0.063-0.200 mm), using chloroform containing 2% of
methanol as eluant, and is collected in 200 cc fractions. The fractions which
are antibiotically active against Aspergillus niger in the plate diffusion test are
combined, evaporated to dryness as described above, and after dissolving in
methanol are chromatographed on 110 g of Sephadex LH20 with the same
solvent, whereupon those 20 cc fractions showing an antibiotic effect against
Aspergillus niger in the test indicated above, are combined. A test in the thin
layer chromatogram, e.g., with silica gel on Polygram foils and
hexane/acetone (1:1) as eluant, indicates that the residue of the methanol
solution evaporated as described above mainly consists of the two new
antibiotics S 7481/F-1 and S 7481/F-2. These are separated and
simultaneously purified by a further chromatography of the mixture thereof,
using a 1,000-fold amount of silica gel on the above indicated quality and
chloroform contains 2% of methanol. A testing of the eluate fractions having a
volume in milliliters which is half as large as the weight of the silica gel in
grams, in the thin layer chromatogram, indicates that the antibiotic S 7481/F-
1 appears first in the eluate, followed by a mixture of the two antibiotics and
finally by homogeneous S748l/F-2.
Further amounts of the two antibiotics may be obtained from the mixture by
repeating chromatography under the same conditions. | | brand name | Gengraf (Abbott); Neoral (Novartis); Restasis (Allergan); Sandimmune (Novartis) [Names previously used: Cyclosporin A; Cyclosporin.]. | | Therapeutic Function | Immunosuppressive | | 一般的な説明 | White prismatic needles (from acetone) or white powder. | | 空気と水の反応 | Slightly water soluble . | | 反応プロフィール | Cyclosporin A is an amide. Amides/imides react with azo and diazo compounds to generate toxic gases. Flammable gases are formed by the reaction of organic amides/imides with strong reducing agents. Amides are very weak bases (weaker than water). Imides are less basic yet and in fact react with strong bases to form salts. That is, they can react as acids. Mixing amides with dehydrating agents such as P2O5 or SOCl2 generates the corresponding nitrile. The combustion of these compounds generates mixed oxides of nitrogen (NOx). | | 健康ハザード | SYMPTOMS: Symptoms of exposure to Cyclosporin A include hepatotoxicity, nephrotoxicity, hyperkalemia, hyperuricemia, convulsions, renal dysfunction, tremor, hirsutism, hypertension, gum hyperplasia, cramps, acne, headache, diarrhea, nausea, vomiting, abdominal discomfort, paresthesia, flushing, leukopenia, lymphoma, sinusitis and gynecomastia. In 2% or less of persons exposed, it has caused allergic reactions, anemia, anorexia, confusion, conjunctivitis, edema, fever, brittle fingernails, gastritis, hearing loss, hiccups, hyperglycemia, muscle pain, peptic ulcer, thrombocytopenia and tinnitus. Rare reactions include anxiety, chest pain, constipation, depression, hair breaking hematuria, joint pain, lethargy, mouth sores, myocardial infarction, night sweats, pancreatitis, pruritus, swallowing difficulty, tingling, upper gastrointestinal bleeding, visual disturbance, weakness and weight loss. It has caused kidney and liver damage. An increased susceptibility to infection may occur. Other symptoms include gastrointestinal disturbance, rashes and angioedema. | | 火災危険 | Flash point data for Cyclosporin A are not available; however, Cyclosporin A is probably combustible. | | Biochem/physiol Actions | Cyclosporin A is a non-polar cyclic oligopeptide produced by the fungus Tolypocladium inflatum. It is a potent immunosuppressive agent, affecting primarily T-lymphocytes. It has been shown to inhibit the functioning of several nuclear proteins involved in T-cell activation at the level of mRNA transcription. It forms a complex with its intracellular receptor cyclophilin, which can then bind to calcineurin, a Ca2+- and calmodulin-dependent protein phosphatase, inhibiting its enzymatic activity. CsA was found to suppress the replication of hepatitis C virus genome in cultured hepatocytes. At concentrations >10 nM, CsA protected isolated hepatocytes against the action of phalloidin. CsA can inhibit IL2 production resulting from T cell activation via Calcineurin inhibition.An extensive list of references has been reported, including a comprehensive review of analytical properties. | | 薬理学 | Cyclosporine has no direct effect on keratinocytes and is not a mitotic inhibitor. Cyclosporine inhibits cytokine release, which results in a decreased recruitment of APCs into the epidermis and decreases immunoreactivity of lesions. Potential long-term side effects preclude cyclosporine’s use in all but very severe and recalcitrant psoriasis. Cyclosporine can be combined with lowdose methotrexate. | | 臨床応用 | Immunosuppressant:
Prophylaxis of solid organ transplant rejection
Nephrotic syndrome
Atopic dermatitis
Psoriasis
Rheumatoid arthritis
Ulcerative colitis | | 副作用 | Cyclosporine’s main side effects, even at low doses, are hypertension and nephrotoxicity. Age, baseline blood pressure, and baseline creatinine levels are predictors of higher risks of side effects. Glomerular filtration rate (GFR) is a more sensitive test than creatinine for evaluating renal function, and a baseline is recommended in any high-risk patient. Longterm treatment with CSA may induce interstitial fibrosis and glomerular sclerosis, with more pronounced changes directly associated with duration of therapy. It should be administered only by dermatologists experienced in its use. | | 安全性プロファイル | Confirmed carcinogen
producing Hodghn's dlsease. Experimental
reproductive effects. Poison by
intraperitoneal and intravenous routes.
Moderately toxic by ingestion. Human
systemic effects by ingestion: increased body
temperature, cyanosis. Mutation data
reported. When heated to decomposition it
emits toxic fumes of NOx. | | 合成 | Cyclosporine A, [R-[R*
,R*
-(E)]]-cyclo-(L-alanyl-D-alanyl-N-methyl-L�leucyl-N-methyl-L-leucyl-N-methyl-L-valyl-3-hydroxy-N,4-dimethyl-L-2-amino-6-
octenoyl-L-α-aminobutyryl-N-methylglycyl-N-methyl-L-leucyl-L-valyl-N-methyl-L-leucine)
(31.2.2), is extracted from a cultural liquid of products of the vital activity of the mush�room Tolypocladium inflatum [14–17], and which is also proposed to obtain synthetically. | | 職業ばく露 | Cyclosporin A is a fungal metabolite;
an amide immunosuppressant drug used in various
surgeries. | | 薬物相互作用 | Potentially hazardous interactions with other drugs
Increased risk of hyperkalaemia with ACE
inhibitors, angiotensin-II antagonists, potassium�sparing diuretics, potassium salts.
Increased risk of nephrotoxicity with
aminoglycosides, amphotericin, co-trimoxazole,
disopyramide, foscarnet, melphalan, NSAIDs,
polymyxins, quinolones, sulphonamides, thiazide
diuretics, trimethoprim and vancomycin.
Increased ciclosporin levels with acetazolamide,
aciclovir, amiodarone, atazanavir, boceprevir,
carvedilol, chloramphenicol, chloroquine, cimetidine,
danazol, diltiazem, doxycycline, famotidine,
fluconazole, fluoxetine, fluvoxamine, fosamprenavir,
glibenclamide, glipizide, grapefruit juice,
hydroxychloroquine, imatinib, indinavir, itraconazole,
ketoconazole, lercanidipine (concentration of both
drugs increased - avoid), macrolides, micafungin,
miconazole, high-dose methylprednisolone,
metoclopramide, metronidazole, muromonab�CD3, nicardipine, posaconazole, progestogens,
propafenone, ritonavir, saquinavir and telaprevir
(concentration of both drugs increased), tacrolimus,
verapamil and voriconazole.
Decreased ciclosporin levels with barbiturates,
bupropion, carbamazepine, efavirenz, fosphenytoin,
griseofulvin, lanreotide, modafinil, octreotide,
pasireotide, phenytoin, primidone, quinine, red
wine, rifampicin, St John’s wort, sulfadiazine,
IV sulfadimidine, sulfasalazine, sulfinpyrazone,terbinafine, ticlopidine and IV trimethoprim and
possibly by oxcarbazepine.
Aliskiren: concentration of aliskiren increased -
avoid.
Ambrisentan: concentration of ambrisentan
increased.
Antibacterials: increased risk of myopathy with
daptomycin - try to avoid concomitant use.
Anticoagulants: concentration of dabigatran and
edoxaban increased - avoid with dabigatran and
reduce dose of edoxaban.
Antidiabetics: may increase repaglinide
concentration, risk of hypoglycaemia.
Antimuscarinics: avoid with darifenacin.
Antivirals: avoid with simeprevir, increased
simeprevir concentration; when starting co�administration with dasubavir and ombitasvir/
paritaprevir/ritonavir, give one fifth of the total daily
dose of ciclosporin once daily. Monitor ciclosporin
levels and adjust dose and/or dosing frequency as
needed.
Basiliximab: may alter ciclosporin levels.
Bosentan: co-administration of ciclosporin and
bosentan is contraindicated. When ciclosporin
and bosentan are co-administered, initial trough
concentrations of bosentan are 30 times higher
than normal. At steady state, trough levels are 3-4
times higher than normal. Blood concentrations of
ciclosporin decreased by 50%.
Calcium-channel blockers: increased nifedipine
concentration and toxicity; amlodipine may increase
ciclosporin concentration by up to 40%.
Cardiac glycosides: increased digoxin concentration
and toxicity.
Caspofungin: caspofungin concentration increased -
monitor LFTs.
Colchicine: risk of myopathy or rhabdomyolysis;
also increased blood-ciclosporin concentrations and
nephrotoxicity - avoid.
Cytotoxics: increased risk of neurotoxicity
with doxorubicin; concentration of epirubicin,
everolimus and idarubicin increased; reduced
excretion of mitoxantrone; increased toxicity with
methotrexate; seizures have been reported in bone
marrow transplant patients taking busulfan and
cyclophosphamide; use crizotinib with caution;
concentration of etoposide possibly increased
(increased risk of toxicity); possible interaction with
docetaxol.
Eltrombopag: exposure reduced by ciclosporin.
Fidaxomicin: avoid concomitant use.
Lenalidomide: concentration of lenalidomide
increased.
Lipid-lowering agents: absorption reduced by
colesevelam, increased risk of myopathy with statins
(avoid with simvastatin, max dose of atorvastatin
should be 10 mg1
); avoid with rosuvastatin; increased
risk of nephrotoxicity with fenofibrate; bezafibrate
may increase creatinine and reduce ciclosporin levels;
concentration of both drugs may be increased with
ezetimibe.
Mycophenolate mofetil: some studies show that
ciclosporin decreases plasma MPA AUC levels - no
dose change required.
NSAIDs: diclofenac concentration increased -
reduce diclofenac dose.
Omeprazole: may alter ciclosporin concentration.
Orlistat: absorption of ciclosporin possibly reduced.
Prednisolone: increased prednisolone concentration.
Rifaximin: concentration of rifaximin increased.
Sirolimus: increased absorption of sirolimus -
give sirolimus 4 hours after ciclosporin; sirolimus
concentration increased; long term concomitant
administration may be associated with deterioration
in renal function.
Tacrolimus: increased ciclosporin concentration and
toxicity - avoid.
Ursodeoxycholic acid: unpredictably increased
absorption and raised ciclosporin levels in some
patients. | | 発がん性 | Cyclosporin A is known to be a human carcinogen based on sufficient evidence of carcinogenicity from studies in humans. | | 代謝 | Ciclosporin is widely distributed throughout the body.
Distribution in the blood is concentration-dependent,
with between 41-58% in erythrocytes and 10-20% in
leucocytes; the remainder is found in plasma, about 90%
protein-bound, mostly to lipoprotein.
Clearance from the blood is biphasic. Ciclosporin is
extensively metabolised in the liver and mainly excreted
in faeces via the bile. About 6% of a dose is reported to be
excreted in the urine, less than 0.1% unchanged. | | 貯蔵 | -20°C (desiccate) | | 輸送方法 | UN3249 Medicine, solid, toxic, n.o.s., Hazard
Class: 6.1; Labels: 6.1-Poisonous materials. | | 不和合性 | Amides/imides react with azo and diazo
compounds to generate toxic gases. Flammable gases are
formed by the reaction of organic amides/imides with
strong reducing agents such as hydrideds and active metals.
Amides are very weak bases (weaker than water). Imides
are less basic yet and in fact react with strong bases to
form salts. That is, they can react as acids. Mixing amides
with dehydrating agents such as such as phosphorus pent-
oxide or thionyl chloride generates the corresponding
nitrile. The combustion of these compounds generates
mixed oxides of nitrogen (NOx). | | 廃棄物の処理 | t is inappropriate and possibly
dangerous to the environment to dispose of expired or waste
drugs and pharmaceuticals by flushing them down the toilet
or discarding them to the trash. Household quantities of
expired or waste pharmaceuticals may be mixed with wet
cat litter or coffee grounds, double-bagged in plastic, discard
in trash. Larger quantities shall carefully take into consider-
ation applicable DEA, EPA, and FDA regulations. If possi-
ble return the pharmaceutical to the manufacturer for proper
disposal being careful to properly label and securely package
the material. Alternatively, the waste pharmaceutical shall be
labeled, securely packaged, and transported by a state
licensed medical waste contractor to dispose by burial in a
licensed hazardous or toxic waste landfill or incinerator. | | 参考文献 | 1) Liu et al. (1993), FK506 and cyclosporin: molecular probes for studying intracellular signal transduction; Trends Pharmacol. Sci., 18 334 |
|