- cupric carbonate
-

- $10.00 / 1KG
-
2021-09-10
- CAS:1184-64-1
- Min. Order: 1KG
- Purity: 99%
- Supply Ability: 10 mt
- cupric carbonate
-

- $1.00 / 1KG
-
2020-01-08
- CAS:1184-64-1
- Min. Order: 1KG
- Purity: 98% HPLC
- Supply Ability: 10 tons/month
|
| Product Name: | cupric carbonate | | Synonyms: | Carbonic acid, copper(2+) salt (1:1);Copper carbonate (CuCo3);Copper(Ⅱ)carbonate monohydrate;Copper oxycarbonate;CARBONICACID,COPPER(II)SALT;cooper carbonate | | CAS: | 1184-64-1 | | MF: | CH2CuO3 | | MW: | 125.57 | | EINECS: | 214-671-4 | | Product Categories: | Inorganics;K00001 | | Mol File: | 1184-64-1.mol | 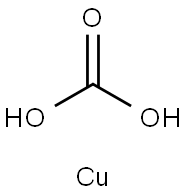 |
| | cupric carbonate Chemical Properties |
| storage temp. | Inert atmosphere,Room Temperature | | solubility | insoluble in H2O | | color | crystals, crystalline | | Water Solubility | insoluble H2O | | Stability: | Stable. Incompatible with strong acids. | | LogP | -0.809 (est) | | EPA Substance Registry System | Cupric carbonate (1184-64-1) |
| | cupric carbonate Usage And Synthesis |
| Description | Copper(II) carbonate or cupric carbonate is a chemical compound with formula CuCO3. At ambient temperatures, it is an ionic solid (a salt) consisting of copper(II) cations Cu2+and carbonate anions CO32− . | | Synthesis | Reliable synthesis of true copper(II) carbonate was reported for the first time in 1973 by Hartmut Ehrhardt and others. The compound was obtained as a gray powder, by heating basic copper carbonate in an atmosphere of carbon dioxide (produced by the decomposition of silver oxalate Ag2C2O4) at 500 °C and 2 GPa (20,000 atm). The compound was determined to have a monoclinic structure. | | Chemical Properties | green or blue powder | | Uses | Copper(I) carbonate is known as cuprous carbonate since copper’s
ion is +1; copper(II) carbonate (Cu2+ + CO3
2- → CuCO3) is known as cupric carbonate, which
is also known as the green copper mineral malachite, used in pigments, as an insecticide, as a
cosmetic astringent, and as a plant fungicide to prevent smut. |
| | cupric carbonate Preparation Products And Raw materials |
|